Cervical spinal hemisection alters phrenic motor neuron glutamatergic mRNA receptor expression
- PMID: 35247372
- PMCID: PMC9090312
- DOI: 10.1016/j.expneurol.2022.114030
Cervical spinal hemisection alters phrenic motor neuron glutamatergic mRNA receptor expression
Abstract
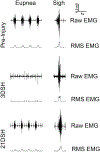
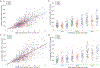
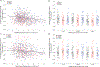
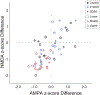
Upper cervical spinal cord injuries (SCI) disrupt descending inputs to phrenic motor neurons (PhMNs), impairing respiratory function. Unilateral spinal hemisection at C2 (C2SH) results in loss of ipsilateral rhythmic diaphragm muscle (DIAm) EMG activity associated with lower force behaviors accomplished by recruitment of smaller PhMNs in rats. Activity during higher force, non-ventilatory behaviors that recruit larger PhMNs is minimally impaired following C2SH. We previously showed neuroplasticity in glutamatergic receptor expression in PhMN post-C2SH with changes in NMDA receptor expression reflecting functional recovery over time. We hypothesize that C2SH-induced changes in glutamatergic receptor (AMPA and NMDA) mRNA expression in PhMNs vary with motor neuron size, with more pronounced changes in smaller PhMNs. Retrogradely-labelled PhMNs were classified in tertiles according to somal surface area and mRNA expression was measured using single-cell, multiplex fluorescence in situ hybridization. Ipsilateral to C2SH, a pronounced reduction in NMDA mRNA expression in PhMNs was evident at 3 days post-injury with similar impact on PhMNs in the lower size tertile (~68% reduction) and upper tertile (~60%); by 21 days, there was near complete restoration of NMDA receptor mRNA expression across all PhMNs. There were no changes in NMDA mRNA expression contralateral to C2SH. There were no changes in AMPA mRNA expression at PhMNs on either side of the spinal cord or at any time-point post-C2SH. In summary, following C2SH there is ipsilateral reduction in PhMN NMDA mRNA expression at 3 days that is not limited to smaller PhMN recruited in the generation of lower force ventilatory behaviors. The recovery of NMDA mRNA expression by 21 days post-C2SH is consistent with evidence of spontaneous recovery of ipsilateral DIAm activity at this timepoint. These findings suggest a possible role for NMDA receptor mediated glutamatergic signaling in mechanisms supporting postsynaptic neuroplasticity at the PhMN pool and recovery of DIAm activity after cervical SCI.
Keywords: Glutamate; Motor unit recruitment; Neuromotor control; Neurotransmitter; Phrenic motor neurons; Respiration.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures
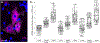

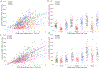
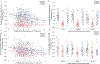
Similar articles
-
Disproportionate loss of excitatory inputs to smaller phrenic motor neurons following cervical spinal hemisection.J Physiol. 2020 Oct;598(20):4693-4711. doi: 10.1113/JP280130. Epub 2020 Aug 19. J Physiol. 2020. PMID: 32735344 Free PMC article.
-
Impact of upper cervical spinal cord hemisection on diaphragm neuromotor control.J Neurophysiol. 2025 Aug 1;134(2):698-714. doi: 10.1152/jn.00279.2025. Epub 2025 Jul 28. J Neurophysiol. 2025. PMID: 40720229 Free PMC article.
-
Heterogeneous glutamatergic receptor mRNA expression across phrenic motor neurons in rats.J Neurochem. 2020 Jun;153(5):586-598. doi: 10.1111/jnc.14881. Epub 2019 Oct 17. J Neurochem. 2020. PMID: 31563147 Free PMC article.
-
Functional recovery after cervical spinal cord injury: Role of neurotrophin and glutamatergic signaling in phrenic motoneurons.Respir Physiol Neurobiol. 2016 Jun;226:128-36. doi: 10.1016/j.resp.2015.10.009. Epub 2015 Oct 23. Respir Physiol Neurobiol. 2016. PMID: 26506253 Free PMC article. Review.
-
The bulbospinal network controlling the phrenic motor system: Laterality and course of descending projections.Neurosci Res. 2017 Aug;121:7-17. doi: 10.1016/j.neures.2017.03.004. Epub 2017 Apr 4. Neurosci Res. 2017. PMID: 28389264 Review.
Cited by
-
Contrasting Experimental Rodent Aftercare With Human Clinical Treatment for Cervical Spinal Cord Injury: Bridging the Translational "Valley of Death".J Neurotrauma. 2023 Dec;40(23-24):2469-2486. doi: 10.1089/neu.2023.0314. Epub 2023 Nov 10. J Neurotrauma. 2023. PMID: 37772694 Free PMC article. Review.
-
An update on spinal cord injury and diaphragm neuromotor control.Expert Rev Respir Med. 2025 Jul;19(7):679-695. doi: 10.1080/17476348.2025.2495165. Epub 2025 Apr 22. Expert Rev Respir Med. 2025. PMID: 40258801 Review.
-
Acute ampakines increase voiding function and coordination in a rat model of SCI.bioRxiv [Preprint]. 2023 Nov 17:2023.05.26.542339. doi: 10.1101/2023.05.26.542339. bioRxiv. 2023. Update in: Elife. 2024 Mar 07;12:RP89767. doi: 10.7554/eLife.89767. PMID: 37293023 Free PMC article. Updated. Preprint.
-
Chemogenetic stimulation of phrenic motor output and diaphragm activity.Elife. 2025 Jun 2;13:RP97846. doi: 10.7554/eLife.97846. Elife. 2025. PMID: 40454577 Free PMC article.
-
AMPK-Nrf2 Signaling Pathway in Phrenic Motoneurons following Cervical Spinal Cord Injury.Antioxidants (Basel). 2022 Aug 26;11(9):1665. doi: 10.3390/antiox11091665. Antioxidants (Basel). 2022. PMID: 36139739 Free PMC article.
References
-
- Chitravanshi VC, Sapru HN, 1996. NMDA as well as non-NMDA receptors mediate the neurotransmission of inspiratory drive to phrenic motoneurons in the adult rat. Brain Res 715, 104–112. - PubMed
-
- Devivo MJ, 2012. Epidemiology of traumatic spinal cord injury: trends and future implications. Spinal Cord 50, 365–372. - PubMed
-
- Dureuil B, Viires N, Cantineau JP, Aubier M, Desmonts JM, 1986. Diaphragmatic contractility after upper abdominal surgery. J Appl Physiol (1985) 61, 1775–1780. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

