In situ generation of a Zwitterionic fluorescent probe for detection of human serum albumin protein
- PMID: 35248557
- PMCID: PMC9018593
- DOI: 10.1016/j.ab.2022.114630
In situ generation of a Zwitterionic fluorescent probe for detection of human serum albumin protein
Abstract
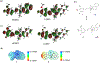
In this article, a new approach for human serum albumin selective fluorophore design has been reported. The fluorophore reported here comprises a substituted phenol donor and a cationic benzo[e]indolium acceptor connected with a π bond. Originally, the cationic fluorophore did not bind with human serum albumin. Upon deprotonation of the phenolic-OH by a water molecule the cationic form was transformed into an active zwitterionic form. Spectroscopic studies and theoretical calculations revealed that the new active form remained in a zwitterionic state in neutral aqueous solution, and it formed a strong supramolecular complex with human serum albumin. The spontaneous complexation resulted multi-fold increase of fluorescence intensity which increased linearly with the concentrations of the protein, thus giving an analytical tool to monitor human serum albumin in aqueous samples. We believe, this simple strategy applied on appropriate fluorogenic scaffolds would prove useful to develop new and improved turn-on fluorescent probes for pH regulated biological applications.
Keywords: Donor-acceptor fluorophore; Human serum albumin; Intramolecular charge transfer; Turn-on fluorescence.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declarations of interest
None
Figures
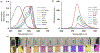
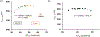
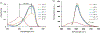
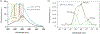
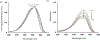
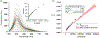
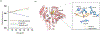
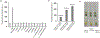
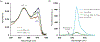
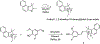
Similar articles
-
Dual luminescent charge transfer probe for quantitative detection of serum albumin in aqueous samples.Spectrochim Acta A Mol Biomol Spectrosc. 2020 Jul 5;235:118305. doi: 10.1016/j.saa.2020.118305. Epub 2020 Mar 24. Spectrochim Acta A Mol Biomol Spectrosc. 2020. PMID: 32259717 Free PMC article.
-
Evaluating the Merit of a Syringol Derived Fluorophore as a Charge Transfer Probe for Detection of Serum Albumins.J Photochem Photobiol A Chem. 2022 Jan 1;422:113563. doi: 10.1016/j.jphotochem.2021.113563. Epub 2021 Sep 30. J Photochem Photobiol A Chem. 2022. PMID: 34720541 Free PMC article.
-
Development of Human Serum Albumin Selective Fluorescent Probe Using Thieno[3,2-b]pyridine-5(4H)-one Fluorophore Derivatives.Sensors (Basel). 2019 Dec 1;19(23):5298. doi: 10.3390/s19235298. Sensors (Basel). 2019. PMID: 31805717 Free PMC article.
-
Devising Efficient Red-Shifting Strategies for Bioimaging: A Generalizable Donor-Acceptor Fluorophore Prototype.Chem Asian J. 2020 May 15;15(10):1514-1523. doi: 10.1002/asia.202000175. Epub 2020 Apr 8. Chem Asian J. 2020. PMID: 32216076 Review.
-
Activity-Based Sensing and Theranostic Probes Based on Photoinduced Electron Transfer.Acc Chem Res. 2019 Oct 15;52(10):2818-2831. doi: 10.1021/acs.accounts.9b00340. Epub 2019 Sep 20. Acc Chem Res. 2019. PMID: 31538473 Review.
Cited by
-
A novel merocyanine photoacid for visible light-controlled pH modulation.J Photochem Photobiol A Chem. 2025 Feb 1;459:116081. doi: 10.1016/j.jphotochem.2024.116081. Epub 2024 Oct 12. J Photochem Photobiol A Chem. 2025. PMID: 39494443
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

