A Biomaterial Model to Assess the Effects of Age in Vascularization
- PMID: 35249009
- PMCID: PMC9440956
- DOI: 10.1159/000523859
A Biomaterial Model to Assess the Effects of Age in Vascularization
Abstract
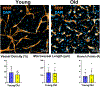
As humans age, there is an increased risk for developing age-associated diseases. Many of these diseases, such as cardiovascular disease, involve dysfunction in the vasculature. Cardiovascular disease stems from endothelial cell dysfunction and reduction in vascularization. Macrophages, prominent innate immune cells involved in orchestrating inflammation and wound healing, have a significant influence on vascularization. While much recent work has investigated the crosstalk between endothelial cells and macrophages, it is still not well defined. The interactions between the cell types are even less understood in specific disease states such as advanced age. Understanding how age influences macrophage/endothelial cell interaction is essential for understanding cardiovascular disease development in the elderly. In the polyethylene glycol (PEG)-based hydrogel system, we model the effects of age on vascularization by encapsulating endothelial cells, pericytes, and human donor macrophages. We created a biomaterial model system in which macrophages, either from young (<35 years old) or old (>65 years old) donors, interact with the modeled vasculature, termed microvessels. Confocal image analysis of vessel density, vessel length, and branch points were used to quantify microvessel growth depending on the age of the macrophage donor. Alongside this, soluble factor secretion and gene expression were evaluated using ELISA and NanoString to showcase biological mechanisms based on the age of each donor. Endothelial cells cultured with macrophages from old donors have reduced microvessel density. There also is reduced soluble factor secretion by the macrophages from old donors, which likely influenced microvessel growth. Altogether, we establish our PEG-based hydrogel vascular model as a system to evaluate patient-specific cell function as well as proposed mechanisms for how age influences microvessels.
Keywords: Aging; Biomaterial; Macrophages; Vasculogenesis.
© 2022 S. Karger AG, Basel.
Conflict of interest statement
Conflict of Interest Statement
No conflict of interest declared
Figures
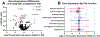


Similar articles
-
3D Printed Pericardium Hydrogels To Promote Wound Healing in Vascular Applications.Biomacromolecules. 2017 Nov 13;18(11):3802-3811. doi: 10.1021/acs.biomac.7b01165. Epub 2017 Oct 16. Biomacromolecules. 2017. PMID: 28976740
-
Vascularized polypeptide hydrogel modulates macrophage polarization for wound healing.Acta Biomater. 2023 Jan 1;155:218-234. doi: 10.1016/j.actbio.2022.11.002. Epub 2022 Nov 15. Acta Biomater. 2023. PMID: 36396041
-
The NG2 Proteoglycan in Pericyte Biology.Adv Exp Med Biol. 2018;1109:5-19. doi: 10.1007/978-3-030-02601-1_2. Adv Exp Med Biol. 2018. PMID: 30523586 Review.
-
Development of a cell-free and growth factor-free hydrogel capable of inducing angiogenesis and innervation after subcutaneous implantation.Acta Biomater. 2019 Nov;99:154-167. doi: 10.1016/j.actbio.2019.08.028. Epub 2019 Aug 16. Acta Biomater. 2019. PMID: 31425892
-
Harnessing Macrophages for Vascularization in Tissue Engineering.Ann Biomed Eng. 2019 Feb;47(2):354-365. doi: 10.1007/s10439-018-02170-4. Epub 2018 Dec 7. Ann Biomed Eng. 2019. PMID: 30535815 Review.
Cited by
-
Metformin Treatment of Macrophages Increases Microvessel Growth in Three-Dimensional Hydrogel Coculture.Tissue Eng Part A. 2024 Aug;30(15-16):460-472. doi: 10.1089/ten.TEA.2023.0327. Epub 2024 Mar 5. Tissue Eng Part A. 2024. PMID: 38308479 Free PMC article.
References
-
- Banarjee R, Sharma A, Bai S, Deshmukh A, Kulkarni M: Proteomic study of endothelial dysfunction induced by AGEs and its possible role in diabetic cardiovascular complications. Journal of Proteomics 2018;187:69–79. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

