Murine Model for Measuring Effects of Humanized-Dosing of Antibiotics on the Gut Microbiome
- PMID: 35250930
- PMCID: PMC8892246
- DOI: 10.3389/fmicb.2022.813849
Murine Model for Measuring Effects of Humanized-Dosing of Antibiotics on the Gut Microbiome
Abstract
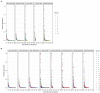
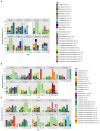
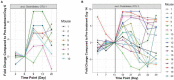
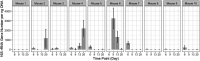
There is a current need for enhancing our insight in the effects of antimicrobial treatment on the composition of human microbiota. Also, the spontaneous restoration of the microbiota after antimicrobial treatment requires better understanding. This is best addressed in well-defined animal models. We here present a model in which immune-competent or neutropenic mice were administered piperacillin-tazobactam (TZP) according to human treatment schedules. Before, during and after the TZP treatment, fecal specimens were longitudinally collected at established intervals over several weeks. Gut microbial taxonomic distribution and abundance were assessed through culture and molecular means during all periods. Non-targeted metabolomics analyses of stool samples using Quadrupole Time of Flight mass spectrometry (QTOF MS) were also applied to determine if a metabolic fingerprint correlated with antibiotic use, immune status, and microbial abundance. TZP treatment led to a 5-10-fold decrease in bacterial fecal viability counts which were not fully restored during post-antibiotic follow up. Two distinct, relatively uniform and reproducible restoration scenarios of microbiota changes were seen in post TZP-treatment mice. Post-antibiotic flora could consist of predominantly Firmicutes or, alternatively, a more diverse mix of taxa. In general, the pre-treatment microbial communities were not fully restored within the screening periods applied. A new species, closely related to Eubacterium siraeum, Mageeibacillus indolicus, and Saccharofermentans acetigenes, became predominant post-treatment in a significant proportion of mice, identified by 16S rRNA gene sequencing. Principal component analysis of QTOF MS of mouse feces successfully distinguished treated from non-treated mice as well as immunocompetent from neutropenic mice. We observe dynamic but distinct and reproducible responses in the mouse gut microbiota during and after TZP treatment and propose the current murine model as a useful tool for defining the more general post-antibiotic effects in the gastro-intestinal ecosystem where humanized antibiotic dosing may ultimately facilitate extrapolation to humans.
Keywords: gastro-intestinal tract; humanized antibiotic treatment; metabolome; microbiome dynamics; non-infected mouse model; piperacillin-tazobactam.
Copyright © 2022 Leopold, Abdelraouf, Nicolau, Agresta, Johnson, Teter, Dunne, Broadwell, van Belkum, Schechter, Sodergren and Weinstock.
Conflict of interest statement
AB, DB, WD, and LS are or were employees of bioMérieux SA (La Balme Les Grottes, France), a company developing, marketing, and selling tests in the infectious disease domain. The company had no influence on the design and execution of the clinical study neither did the company influence the choice of the diagnostic tools used during the clinical study. The opinions expressed in the manuscript are the author’s which do not necessarily reflect company policies. KA and DN have no conflicts to declare.
Figures





References
-
- Bulik C. C., Tessier P. R., Keel R. A., Sutherland C. A., Nicolau D. P. (2012). In vivo comparison of CXA-101 (FR264205) with and without tazobactam versus piperacillin-tazobactam using human simulated exposures against phenotypically diverse gram-negative organisms. Antimicrob. Agents Chemother. 56, 544–549. doi: 10.1128/AAC.01752-10, PMID: - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

