Model-assisted analyses of longitudinal, ordinal outcomes with absorbing states
- PMID: 35253265
- PMCID: PMC9232888
- DOI: 10.1002/sim.9366
Model-assisted analyses of longitudinal, ordinal outcomes with absorbing states
Abstract
Studies of critically ill, hospitalized patients often follow participants and characterize daily health status using an ordinal outcome variable. Statistically, longitudinal proportional odds models are a natural choice in these settings since such models can parsimoniously summarize differences across patient groups and over time. However, when one or more of the outcome states is absorbing, the proportional odds assumption for the follow-up time parameter will likely be violated, and more flexible longitudinal models are needed. Motivated by the VIOLET Study (Ginde et al), a parallel-arm, randomized clinical trial of Vitamin in critically ill patients, we discuss and contrast several treatment effect estimands based on time-dependent odds ratio parameters, and we detail contemporary modeling approaches. In VIOLET, the outcome is a four-level ordinal variable where the lowest "not alive" state is absorbing and the highest "at-home" state is nearly absorbing. We discuss flexible extensions of the proportional odds model for longitudinal data that can be used for either model-based inference, where the odds ratio estimator is taken directly from the model fit, or for model-assisted inferences, where heterogeneity across cumulative log odds dichotomizations is modeled and results are summarized to obtain an overall odds ratio estimator. We focus on direct estimation of cumulative probability model (CPM) parameters using likelihood-based analysis procedures that naturally handle absorbing states. We illustrate the modeling procedures, the relative precision of model-based and model-assisted estimators, and the possible differences in the values for which the estimators are consistent through simulations and analysis of the VIOLET Study data.
Keywords: absorbing state; longitudinal data; marginalized models; ordinal responses; partial proportional odds; proportional odds; randomized clinical trial.
© 2022 John Wiley & Sons Ltd.
Conflict of interest statement
CONFLICT OF INTEREST
None declared.
Figures

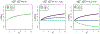

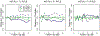

References
-
- McCullagh P. Regression Models for Ordinal Data. Journal of the Royal Statistical Society Series B - Methodological 1980; 42(2): 109–142.
-
- Peterson B, Harrell F. Partial Proportional Odds Models for Ordinal Response Variables. Journal of the Royal Statistical Society Series C-Applied Statistics 1990; 39(2): 205–217.
-
- Frison L, Pocock SJ. Repeated measures in clinical trials: analysis using mean summary statistics and its implications for design. Statistics in Medicine 1992; 11(13): 1685–1704. - PubMed
-
- Yang L, Tsiatis AA. Efficiency study of estimators for a treatment effect in a pretest–posttest trial. The American Statistician 2001; 55(4): 314–321.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
