Regulation of Hepatic Gluconeogenesis by Nuclear Receptor Coactivator 6
- PMID: 35258009
- PMCID: PMC9001147
- DOI: 10.14348/molcells.2022.2222
Regulation of Hepatic Gluconeogenesis by Nuclear Receptor Coactivator 6
Abstract
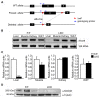
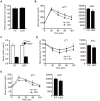
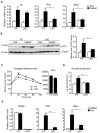
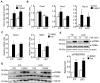
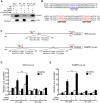
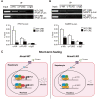
Nuclear receptor coactivator 6 (NCOA6) is a transcriptional coactivator of nuclear receptors and other transcription factors. A general Ncoa6 knockout mouse was previously shown to be embryonic lethal, but we here generated liver-specific Ncoa6 knockout (Ncoa6 LKO) mice to investigate the metabolic function of NCOA6 in the liver. These Ncoa6 LKO mice exhibited similar blood glucose and insulin levels to wild type but showed improvements in glucose tolerance, insulin sensitivity, and pyruvate tolerance. The decrease in glucose production from pyruvate in these LKO mice was consistent with the abrogation of the fasting-stimulated induction of gluconeogenic genes, phosphoenolpyruvate carboxykinase 1 (Pck1) and glucose-6-phosphatase (G6pc). The forskolin-stimulated inductions of Pck1 and G6pc were also dramatically reduced in primary hepatocytes isolated from Ncoa6 LKO mice, whereas the expression levels of other gluconeogenic gene regulators, including cAMP response element binding protein (Creb), forkhead box protein O1 and peroxisome proliferator-activated receptor γ coactivator 1α, were unaltered in the LKO mouse livers. CREB phosphorylation via fasting or forskolin stimulation was normal in the livers and primary hepatocytes of the LKO mice. Notably, it was observed that CREB interacts with NCOA6. The transcriptional activity of CREB was found to be enhanced by NCOA6 in the context of Pck1 and G6pc promoters. NCOA6-dependent augmentation was abolished in cAMP response element (CRE) mutant promoters of the Pck1 and G6pc genes. Our present results suggest that NCOA6 regulates hepatic gluconeogenesis by modulating glucagon/cAMP-dependent gluconeogenic gene transcription through an interaction with CREB.
Keywords: cAMP response element-binding protein; gluconeogenesis; glucose-6-phosphatase; nuclear receptor coactivator 6; phosphoenolpyruvate carboxykinase.
Conflict of interest statement
The authors have no potential conflicts of interest to disclose.
Figures
Similar articles
-
The SMILE transcriptional corepressor inhibits cAMP response element-binding protein (CREB)-mediated transactivation of gluconeogenic genes.J Biol Chem. 2018 Aug 24;293(34):13125-13133. doi: 10.1074/jbc.RA118.002196. Epub 2018 Jun 27. J Biol Chem. 2018. PMID: 29950523 Free PMC article.
-
CREB-upregulated lncRNA MEG3 promotes hepatic gluconeogenesis by regulating miR-302a-3p-CRTC2 axis.J Cell Biochem. 2019 Mar;120(3):4192-4202. doi: 10.1002/jcb.27706. Epub 2018 Sep 27. J Cell Biochem. 2019. PMID: 30260029
-
Regulation of hepatic gluconeogenesis by nuclear factor Y transcription factor in mice.J Biol Chem. 2018 May 18;293(20):7894-7904. doi: 10.1074/jbc.RA117.000508. Epub 2018 Mar 12. J Biol Chem. 2018. PMID: 29530977 Free PMC article.
-
Transcription factors and coactivators controlling nutrient and hormonal regulation of hepatic gluconeogenesis.Int J Biochem Cell Biol. 2012 Jan;44(1):33-45. doi: 10.1016/j.biocel.2011.10.001. Epub 2011 Oct 8. Int J Biochem Cell Biol. 2012. PMID: 22004992 Review.
-
The neglected PCK1/glucagon (inter)action in nutrient homeostasis beyond gluconeogenesis: Disease pathogenesis and treatment.Mol Metab. 2025 Apr;94:102112. doi: 10.1016/j.molmet.2025.102112. Epub 2025 Feb 13. Mol Metab. 2025. PMID: 39954782 Free PMC article. Review.
Cited by
-
The Pleiotropic Face of CREB Family Transcription Factors.Mol Cells. 2023 Jul 31;46(7):399-413. doi: 10.14348/molcells.2023.2193. Epub 2023 Apr 4. Mol Cells. 2023. PMID: 37013623 Free PMC article. Review.
-
Genomic Diversity and Selection Signatures for Zaosheng Cattle.Biology (Basel). 2025 May 28;14(6):623. doi: 10.3390/biology14060623. Biology (Basel). 2025. PMID: 40563874 Free PMC article.
-
Effects of tomato ketchup and tomato paste extract on hepatic lipid accumulation and adipogenesis.Food Sci Biotechnol. 2023 Feb 6;32(8):1111-1122. doi: 10.1007/s10068-023-01244-x. eCollection 2023 Jul. Food Sci Biotechnol. 2023. PMID: 37215254 Free PMC article.
-
Nuclear receptor coactivator 6 is a critical regulator of NLRP3 inflammasome activation and gouty arthritis.Cell Mol Immunol. 2024 Mar;21(3):227-244. doi: 10.1038/s41423-023-01121-x. Epub 2024 Jan 10. Cell Mol Immunol. 2024. PMID: 38195836 Free PMC article.
-
Illuminating Genetic Diversity and Selection Signatures in Matou Goats through Whole-Genome Sequencing Analysis.Genes (Basel). 2024 Jul 12;15(7):909. doi: 10.3390/genes15070909. Genes (Basel). 2024. PMID: 39062688 Free PMC article.
References
-
- Antonson P., Schuster G.U., Wang L., Rozell B., Holter E., Flodby P., Treuter E., Holmgren L., Gustafsson J.Å. Inactivation of the nuclear receptor coactivator RAP250 in mice results in placental vascular dysfunction. Mol. Cell. Biol. 2003;23:1260–1268. doi: 10.1128/MCB.23.4.1260-1268.2003. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

