Targeting ferroptosis suppresses osteocyte glucolipotoxicity and alleviates diabetic osteoporosis
- PMID: 35260560
- PMCID: PMC8904790
- DOI: 10.1038/s41413-022-00198-w
Targeting ferroptosis suppresses osteocyte glucolipotoxicity and alleviates diabetic osteoporosis
Abstract
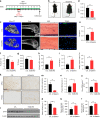
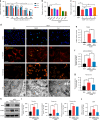
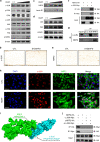
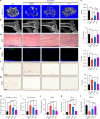
Diabetic osteoporosis (DOP) is the leading complication continuously threatening the bone health of patients with diabetes. A key pathogenic factor in DOP is loss of osteocyte viability. However, the mechanism of osteocyte death remains unclear. Here, we identified ferroptosis, which is iron-dependent programmed cell death, as a critical mechanism of osteocyte death in murine models of DOP. The diabetic microenvironment significantly enhanced osteocyte ferroptosis in vitro, as shown by the substantial lipid peroxidation, iron overload, and aberrant activation of the ferroptosis pathway. RNA sequencing showed that heme oxygenase-1 (HO-1) expression was notably upregulated in ferroptotic osteocytes. Further findings revealed that HO-1 was essential for osteocyte ferroptosis in DOP and that its promoter activity was controlled by the interaction between the upstream NRF2 and c-JUN transcription factors. Targeting ferroptosis or HO-1 efficiently rescued osteocyte death in DOP by disrupting the vicious cycle between lipid peroxidation and HO-1 activation, eventually ameliorating trabecular deterioration. Our study provides insight into DOP pathogenesis, and our results provide a mechanism-based strategy for clinical DOP treatment.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Napoli N, et al. Mechanisms of diabetes mellitus-induced bone fragility. Nat. Rev. Endocrinol. 2017;13:208–219. - PubMed
-
- Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos. Int. 2006;17:1726–1733. - PubMed
-
- Shanbhogue VV, Hansen S, Frost M, Brixen K, Hermann AP. Bone disease in diabetes: another manifestation of microvascular disease? Lancet Diabetes Endocrinol. 2017;5:827–838. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

