Rationale and Design of the COVID-19 Outpatient Prevention Evaluation (COPE - Coalition V) Randomized Clinical Trial: Hydroxychloroquine vs. Placebo in Non-Hospitalized Patients
- PMID: 35262569
- PMCID: PMC8856682
- DOI: 10.36660/abc.20210832
Rationale and Design of the COVID-19 Outpatient Prevention Evaluation (COPE - Coalition V) Randomized Clinical Trial: Hydroxychloroquine vs. Placebo in Non-Hospitalized Patients
Abstract
Background: Despite the need for targeting specific therapeutic options for coronavirus disease 2019 (COVID-19), there has been no evidence of effectiveness of any specific treatment for the outpatient clinical setting. There are few randomized studies evaluating hydroxychloroquine (HCQ) in non-hospitalized patients. These studies indicate no benefit from the use of HCQ, but they assessed different primary outcomes and presented important biases for outcome evaluation.
Objective: To evaluate if HCQ may prevent hospitalization due to COVID-19 compared to a matching placebo.
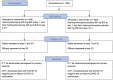
Methods: The COVID-19 Outpatient Prevention Evaluation (COPE) study is a pragmatic, randomized, double-blind, placebo-controlled clinical trial evaluating the use of HCQ (800 mg on day 1 and 400 mg from day 2 to day 7) or matching placebo for the prevention of hospitalization due to COVID-19 in early non-hospitalized confirmed or suspected cases. Inclusion criteria are adults (≥ 18 years) seeking medical care with mild symptoms of COVID-19, with randomization ≤ 7 days after symptom onset, without indication of hospitalization at study screening, and with at least one risk factor for complication (> 65 years; hypertension; diabetes mellitus; asthma; chronic obstructive pulmonary disease or other chronic lung diseases; smoking; immunosuppression; or obesity). All hypothesis tests will be two-sided. A p-value < 0.05 will be considered statistically significant in all analyses. Clinicaltrials.gov: NCT04466540.
Results: Clinical outcomes will be centrally adjudicated by an independent clinical event committee blinded to the assigned treatment groups. The primary efficacy endpoint will be assessed following the intention-to-treat principle.
Conclusion: This study has the potential to reliably answer the scientific question of HCQ use in outpatients with COVID-19. To our knowledge, this is the largest trial evaluating HCQ in non-hospitalized individuals with COVID-19.
Fundamento: Apesar da necessidade de opções terapêuticas específicas para a doença do coronavírus 2019 (covid-19), ainda não há evidências da eficácia de tratamentos específicos no contexto ambulatorial. Há poucos estudos randomizados que avaliam a hidroxicloroquina (HCQ) em pacientes não hospitalizados. Esses estudos não indicaram benefício com o uso da HCQ; no entanto, avaliaram desfechos primários diferentes e apresentaram vieses importantes na avaliação dos desfechos.
Objetivo: Investigar se a HCQ possui o potencial de prevenir hospitalizações por covid-19 quando comparada ao placebo correspondente.
Métodos: O estudo COVID-19 Outpatient Prevention Evaluation (COPE) é um ensaio clínico randomizado, pragmático, duplo-cego, multicêntrico e controlado por placebo que avalia o uso da HCQ (800 mg no dia 1 e 400 mg do dia 2 ao dia 7) ou placebo correspondente na prevenção de hospitalizações por covid-19 em casos precoces confirmados ou suspeitos de pacientes não hospitalizados. Os critérios de inclusão são adultos (≥ 18 anos) que procuraram atendimento médico com sintomas leves de covid-19, com randomização ≤ 7 dias após o início dos sintomas, sem indicação de hospitalização na triagem do estudo e com pelo menos um fator de risco para complicações (> 65 anos, hipertensão, diabetes melito, asma, doença pulmonar obstrutiva crônica ou outras doenças pulmonares crônicas, tabagismo, imunossupressão ou obesidade). Todos os testes de hipótese serão bilaterais. Um valor de p < 0,05 será considerado estatisticamente significativo em todas as análises. Clinicaltrials.gov: NCT04466540.
Resultados: Os desfechos clínicos serão avaliados centralmente por um comitê de eventos clínicos independente cegado para a alocação dos grupos de tratamento. O desfecho primário de eficácia será avaliado de acordo com o princípio da intenção de tratar.
Conclusão: Este estudo apresenta o potencial de responder de forma confiável a questão científica do uso da HCQ em pacientes ambulatoriais com covid-19. Do nosso conhecimento, este é o maior estudo avaliando o uso de HCQ em indivíduos com covid-19 não hospitalizados.
Figures



References
-
- World Health Organization. Clinical Management of Severe Acute Respiratory Infection When Novel Coronavirus (nCoV) Infection is Suspected. Geneva: WHO Library; 2020.
-
- Abella BS, Jolkovsky EL, Biney BT, Uspal JE, Hyman MC, Frank I, et al. Efficacy and Safety of Hydroxychloroquine vs Placebo for Pre-Exposure SARS-CoV-2 Prophylaxis Among Health Care Workers: A Randomized Clinical Trial. JAMA Intern Med. 2021;181(2):195-202. doi: 10.1001/jamainternmed.2020.6319. - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

