Heterogeneous Trimetallic Nanoparticles as Catalysts
- PMID: 35263103
- PMCID: PMC8949769
- DOI: 10.1021/acs.chemrev.1c00493
Heterogeneous Trimetallic Nanoparticles as Catalysts
Abstract
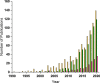
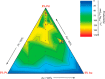
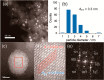
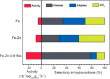
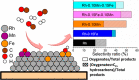
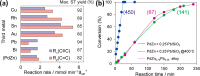
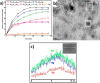
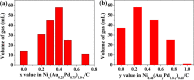
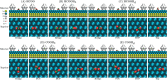
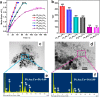
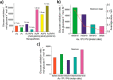
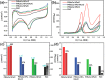
The development and application of trimetallic nanoparticles continues to accelerate rapidly as a result of advances in materials design, synthetic control, and reaction characterization. Following the technological successes of multicomponent materials in automotive exhausts and photovoltaics, synergistic effects are now accessible through the careful preparation of multielement particles, presenting exciting opportunities in the field of catalysis. In this review, we explore the methods currently used in the design, synthesis, analysis, and application of trimetallic nanoparticles across both the experimental and computational realms and provide a critical perspective on the emergent field of trimetallic nanocatalysts. Trimetallic nanoparticles are typically supported on high-surface-area metal oxides for catalytic applications, synthesized via preparative conditions that are comparable to those applied for mono- and bimetallic nanoparticles. However, controlled elemental segregation and subsequent characterization remain challenging because of the heterogeneous nature of the systems. The multielement composition exhibits beneficial synergy for important oxidation, dehydrogenation, and hydrogenation reactions; in some cases, this is realized through higher selectivity, while activity improvements are also observed. However, challenges related to identifying and harnessing influential characteristics for maximum productivity remain. Computation provides support for the experimental endeavors, for example in electrocatalysis, and a clear need is identified for the marriage of simulation, with respect to both combinatorial element screening and optimal reaction design, to experiment in order to maximize productivity from this nascent field. Clear challenges remain with respect to identifying, making, and applying trimetallic catalysts efficiently, but the foundations are now visible, and the outlook is strong for this exciting chemical field.
Conflict of interest statement
The authors declare no competing financial interest.
Figures












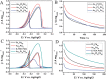
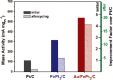

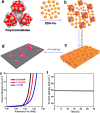
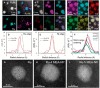
References
-
- Chalmin E.; Menu M.; Vignaud C. Analysis of Rock Art Painting and Technology of Palaeolithic Painters. Meas. Sci. Technol. 2003, 14, 1590–1597. 10.1088/0957-0233/14/9/310. - DOI
-
- Mohan Bhagyaraj S.; Oluwafemi O. S.. Nanotechnology: The Science of the Invisible. In Synthesis of Inorganic Nanomaterials; Elsevier, 2018; pp 1–18. 10.1016/B978-0-08-101975-7.00001-4. - DOI
-
- Brun N.; Mazerolles L.; Pernot M. Microstructure of Opaque Red Glass Containing Copper. J. Mater. Sci. Lett. 1991, 10, 1418–1420. 10.1007/BF00735696. - DOI
-
- Khawaji M.; Chadwick D. Selective Catalytic Oxidation over Au-Pd/Titanate Nanotubes and the Influence of the Catalyst Preparation Method on the Activity. Catal. Today 2019, 334, 122–130. 10.1016/j.cattod.2018.11.080. - DOI
-
- Sun Y.; Mayers B.; Xia Y. Metal Nanostructures with Hollow Interiors. Adv. Mater. 2003, 15, 641–646. 10.1002/adma.200301639. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

