TRACEBACK: Testing of Historical Tubo-Ovarian Cancer Patients for Hereditary Risk Genes as a Cancer Prevention Strategy in Family Members
- PMID: 35263119
- PMCID: PMC9197360
- DOI: 10.1200/JCO.21.02108
TRACEBACK: Testing of Historical Tubo-Ovarian Cancer Patients for Hereditary Risk Genes as a Cancer Prevention Strategy in Family Members
Abstract
Purpose: Tubo-ovarian cancer (TOC) is a sentinel cancer for BRCA1 and BRCA2 pathogenic variants (PVs). Identification of a PV in the first member of a family at increased genetic risk (the proband) provides opportunities for cancer prevention in other at-risk family members. Although Australian testing rates are now high, PVs in patients with TOC whose diagnosis predated revised testing guidelines might have been missed. We assessed the feasibility of detecting PVs in this population to enable genetic risk reduction in relatives.
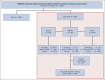
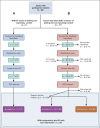
Patients and methods: In this pilot study, deceased probands were ascertained from research cohort studies, identification by a relative, and gynecologic oncology clinics. DNA was extracted from archival tissue or stored blood for panel sequencing of 10 risk-associated genes. Testing of deceased probands ascertained through clinic records was performed with a consent waiver.
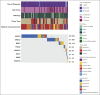
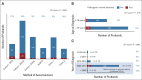
Results: We identified 85 PVs in 84 of 787 (11%) probands. Familial contacts of 39 of 60 (65%) deceased probands with an identified recipient (60 of 84; 71%) have received a written notification of results, with follow-up verbal contact made in 85% (33 of 39). A minority of families (n = 4) were already aware of the PV. For many (29 of 33; 88%), the genetic result provided new information and referral to a genetic service was accepted in most cases (66%; 19 of 29). Those who declined referral (4 of 29) were all male next of kin whose family member had died more than 10 years before.
Conclusion: We overcame ethical and logistic challenges to demonstrate that retrospective genetic testing to identify PVs in previously untested deceased probands with TOC is feasible. Understanding reasons for a family member's decision to accept or decline a referral will be important for guiding future TRACEBACK projects.
Conflict of interest statement
Figures
Comment in
-
Challenges for clinical application of "TRACEBACK" study: testing of historical Tubo-Ovarian cancer patients for hereditary risk genes.Ann Transl Med. 2023 Apr 28;11(8):295. doi: 10.21037/atm-23-352. Epub 2023 Feb 23. Ann Transl Med. 2023. PMID: 37181337 Free PMC article. No abstract available.
References
-
- Kuchenbaecker KB, Hopper JL, Barnes DR, et al. : Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA 317:2402-2416, 2017 - PubMed
-
- Rafnar T, Gudbjartsson DF, Sulem P, et al. : Mutations in BRIP1 confer high risk of ovarian cancer. Nat Genet 43:1104-1107, 2011 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

