The Effect of Cutibacterium acnes Infection on Nerve Penetration in the Annulus Fibrosus of Lumbar Intervertebral Discs via Suppressing Oxidative Stress
- PMID: 35265268
- PMCID: PMC8898795
- DOI: 10.1155/2022/9120674
The Effect of Cutibacterium acnes Infection on Nerve Penetration in the Annulus Fibrosus of Lumbar Intervertebral Discs via Suppressing Oxidative Stress
Abstract
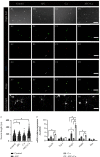
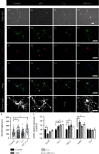
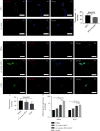
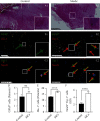
Modic changes (MCs) and low back pain are highly correlated and an economic burden to the society. Previous studies have shown that Cutibacterium acnes (C. acnes) infection can lead to MCs. The purpose of this study was to clarify whether and how C. acnes contributes to oxidative stress and nerve growth that potentially leads to low back pain. Neurons from the hippocampus or dorsal root ganglion (DRG) of Sprague-Dawley (SD) rats were cocultured with annulus fibrosus cells (AFCs) with or without the presence of the C. acnes supernatant in vitro. Cell viability, neurite length, oxidative stress, and neuro-related gene expression were examined. Furthermore, samples from the patients with MCs and SD rat model of MCs were used to validate the nerve growth results. Neurons from both the hippocampus and DRG showed neurites when cocultured with AFCs in the environment with/without the C. acnes supernatant. The average neurite length was significantly longer when exposed to the C. acnes supernatant in the hippocampal neuron (217.1 ± 90.0 μm versus 150.1 ± 68.1 μm in the control group) and in the DRG neuron (229.1 ± 91.3 μm versus 149.2 ± 64.8 μm in the control group). Hippocampal neurons showed upregulated expression levels of NeuN, Map2, and Psd95, while upregulation was only seen in Tuj-1 in DRG neurons. Suppressed oxidative stress could be observed using axon growth symbols. Degenerated disc structures and abnormal bone remodelling were found in animal models and clinical samples of MCs, with astrocytes, microglia, and neurons in the disc. Therefore, C. acnes infection was found to cause back pain in the presence of MCs by promoting nerve penetration into the annulus fibrosus by suppressing oxidative stress.
Copyright © 2022 Zhi Shan et al.
Conflict of interest statement
The authors declare that there is no conflict of interest regarding the publication of this paper.
Figures

Similar articles
-
Lumbar posterolateral fusion inhibits sensory nerve ingrowth into punctured lumbar intervertebral discs and upregulation of CGRP immunoreactive DRG neuron innervating punctured discs in rats.Eur Spine J. 2010 Apr;19(4):593-600. doi: 10.1007/s00586-009-1237-9. Epub 2009 Dec 12. Eur Spine J. 2010. PMID: 20012755 Free PMC article.
-
Existence of nerve growth factor receptors, tyrosine kinase a and p75 neurotrophin receptors in intervertebral discs and on dorsal root ganglion neurons innervating intervertebral discs in rats.Spine (Phila Pa 1976). 2008 Sep 1;33(19):2047-51. doi: 10.1097/BRS.0b013e31817f8d58. Spine (Phila Pa 1976). 2008. PMID: 18758359
-
Propionibacterium acnes Incubation in the Discs Can Result in Time-Dependent Modic Changes: A Long-Term Rabbit Model.Spine (Phila Pa 1976). 2017 Nov 1;42(21):1595-1603. doi: 10.1097/BRS.0000000000002192. Spine (Phila Pa 1976). 2017. PMID: 28399545
-
The role of Propionibacterium acnes in and Modic type 1 changes : A literature review.J Med Invest. 2020;67(1.2):21-26. doi: 10.2152/jmi.67.21. J Med Invest. 2020. PMID: 32378611 Review.
-
Overview: the role of Propionibacterium acnes in nonpyogenic intervertebral discs.Int Orthop. 2016 Jun;40(6):1291-8. doi: 10.1007/s00264-016-3115-5. Epub 2016 Jan 28. Int Orthop. 2016. PMID: 26820744 Review.
Cited by
-
New-Occurrence of Postoperative Modic Changes and Its Influence on the Surgical Prognosis After Percutaneous Endoscopic Lumbar Disc Discectomy.Orthop Surg. 2025 Feb;17(2):482-491. doi: 10.1111/os.14308. Epub 2024 Dec 5. Orthop Surg. 2025. PMID: 39639493 Free PMC article.
-
Induction of cervical disc degeneration and discogenic pain by low concentration Propionibacterium acnes infection: an in vivo animal study.Arthritis Res Ther. 2024 Jan 31;26(1):41. doi: 10.1186/s13075-024-03269-x. Arthritis Res Ther. 2024. PMID: 38297365 Free PMC article.
-
Would Cutibacterium acnes Be the Villain for the Chronicity of Low Back Pain in Degenerative Disc Disease? Preliminary Results of an Analytical Cohort.J Pers Med. 2023 Mar 29;13(4):598. doi: 10.3390/jpm13040598. J Pers Med. 2023. PMID: 37108984 Free PMC article.
References
MeSH terms
Supplementary concepts
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

