Traumatic injury clinical trial evaluating tranexamic acid in children (TIC-TOC): A pilot randomized trial
- PMID: 35266589
- PMCID: PMC9463410
- DOI: 10.1111/acem.14481
Traumatic injury clinical trial evaluating tranexamic acid in children (TIC-TOC): A pilot randomized trial
Abstract
Background: The antifibrinolytic drug tranexamic acid (TXA) improves survival in adults with traumatic hemorrhage; however, the drug has not been evaluated in a trial in injured children. We assessed the feasibility of a large-scale trial evaluating the effects of TXA in children with severe hemorrhagic injuries.
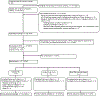
Methods: Severely injured children (0 up to 18th birthday) were randomized into a double-blind randomized trial of (1) TXA 15 mg/kg bolus dose, followed by 2 mg/kg/h infusion over 8 h, (2) TXA 30 mg/kg bolus dose, followed by 4 mg/kg/h infusion over 8 h, or (3) normal saline placebo bolus and infusion. The trial was conducted at four pediatric Level I trauma centers in the United States between June 2018 and March 2020. We enrolled patients under federal exception from informed consent (EFIC) procedures when parents were unable to provide informed consent. Feasibility outcomes included the rate of enrollment, adherence to intervention arms, and ability to measure the primary clinical outcome. Clinical outcomes included global functioning (primary), working memory, total amount of blood products transfused, intracranial hemorrhage progression, and adverse events. The target enrollment rate was at least 1.25 patients per site per month.
Results: A total of 31 patients were randomized with a mean age of 10.7 years (standard deviation [SD] 5.0 years) and 22 (71%) patients were male. The mean time from injury to randomization was 2.4 h (SD 0.6 h). Sixteen (52%) patients had isolated brain injuries and 15 (48%) patients had isolated torso injuries. The enrollment rate using EFIC was 1.34 patients per site per month. All eligible enrolled patients received study intervention (nine patients TXA 15 mg/kg bolus dose, 10 patients TXA 30 mg/kg bolus dose, and 12 patients placebo) and had the primary outcome measured. No statistically significant differences in any of the clinical outcomes were identified.
Conclusion: Based on enrollment rate, protocol adherence, and measurement of the primary outcome in this pilot trial, we confirmed the feasibility of conducting a large-scale, randomized trial evaluating the efficacy of TXA in severely injured children with hemorrhagic brain and/or torso injuries using EFIC.
Keywords: clinical trial; pediatric trauma; tranexamic acid.
© 2022 Society for Academic Emergency Medicine.
Conflict of interest statement
Disclosures:
The authors declare that they have no competing interests currently.
References
-
- 10 Leading Causes of Death by Age Group, United States - 2010. Centers for Disease Control and Prevention; (2010). Accessed November 1, 2021 at: http://www.cdc.gov/injury/wisqars/pdf/10lcid_all_deaths_by_age_group_201....
-
- Camara-Costa H, Francillette L, Opatowski M, et al. Self- and Parent-Reported Fatigue 7 Years After Severe Childhood Traumatic Brain Injury: Results of the Traumatisme Grave de l’Enfant Prospective Longitudinal Study. J Head Trauma Rehabil. 2020;35(2):104–116. - PubMed
Grants and funding
- L30 TR000286/TR/NCATS NIH HHS/United States
- MR/M009211/1/MRC_/Medical Research Council/United Kingdom
- University of Utah
- R34 HL135214/HL/NHLBI NIH HHS/United States
- HRSA/HRSA HHS/United States
- U24TR001597/TR/NCATS NIH HHS/United States
- U.S. Department of Health and Human Services
- University of California
- NH/NIH HHS/United States
- U03MC00001/Maternal and Child Health Bureau
- R34HL135214/HL/NHLBI NIH HHS/United States
- U03MC39944/Maternal and Child Health Bureau
- U24 TR001597/TR/NCATS NIH HHS/United States