The Characteristics and Distribution of α2D-, α2B- and α2C-Adrenoceptor Subtypes in Goats
- PMID: 35268232
- PMCID: PMC8909296
- DOI: 10.3390/ani12050664
The Characteristics and Distribution of α2D-, α2B- and α2C-Adrenoceptor Subtypes in Goats
Abstract
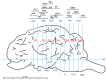
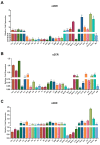
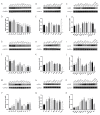
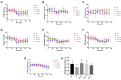
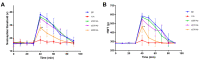
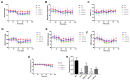
α2-Adrenegic receptors (α2Rs) are important presynaptic modulators of central noradrenergic function (auto receptors) and postsynaptic mediators of many of the widespread effects of catecholamines and related drugs. Studies have shown that ruminants (such as goats and cattle) express special α2DR subtypes in addition to α2BR and α2CR. Real-time quantitative PCR and Western blotting were used to investigate the distribution and density of α2R in different nuclei of the goat central nervous system, selected regions of the spinal cord (L4-L6), and in various peripheral tissues. α2-AR subtype-specific antibodies were injected intrathecally and intracerebroventricularly into the tested goats to block the corresponding subtype of receptors. Pain threshold and physiological parameters were evaluated to explore the functional characteristics of α2BR, α2CR and α2DR in goats. Our results suggest that the expression of the mRNAs and proteins of all three α2R subtypes are widely but unevenly distributed in the goat CNS and peripheral tissues. Furthermore, α2DR plays a more important role in α2R-mediated analgesia in goats than α2BR and α2CR, whereas α2CR activation exerts a greater effect on body temperature than α2BR and α2DR.
Keywords: analgesia; goat; intracerebroventricular injection; intrathecal injection; α2 adrenoceptor subtypes.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
-
- Bantel C., Maze M., Stone L., Wilcox G. Encyclopedia of Pain. Springer; Berlin/Heidelberg, Germany: 2007. Alpha(α) 2-Adrenergic Agonists in Pain Treatment; pp. 58–61.
-
- McKelvey D., Hollingshead K.W. Veterinary Anesthesia and Analgesia. 3rd ed. Mosby; St Louis, MO, USA: 2003.
-
- Shah Z., Ding M.-X., Hu M.-L. A review on the current use of alpha2 agonists in small ruminants. Kafkas Univ. Vet. Fak. Derg. 2014;20:633–639.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

