Early Access to Medicines: Use of Multicriteria Decision Analysis (MCDA) as a Decision Tool in Catalonia (Spain)
- PMID: 35268443
- PMCID: PMC8910942
- DOI: 10.3390/jcm11051353
Early Access to Medicines: Use of Multicriteria Decision Analysis (MCDA) as a Decision Tool in Catalonia (Spain)
Abstract
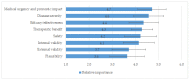
Early access to medicines allows the prescription of a medicine before it is available in the public formulary to patients with severe or rare diseases with high unmet needs who have no authorised therapeutic alternatives available. In this context, consistent decision making is difficult, and a systematic assessment procedure could be useful to tackle complex situations and guarantee the equity of medicines' access. A multidisciplinary panel (MP) conducted four workshops to develop an early access framework based on a reflective multiple criteria decision analysis (MCDA). A set of 12 criteria was agreed: eight quantitative (severity of disease, urgency, efficacy, safety, internal and external validity, therapeutic benefit and plausibility) and four qualitative (therapeutic alternative, existence of precedents, management impact and costs). Quantitative criteria were weighted using a five-point scale. The relative importance of quantitative criteria had mean weights from 4.7 to 3.6, showing its relevance in the decisions. The framework was tested using two case studies, and reliability was assessed by re-test. The re-test revealed no statistical differences, indicating the consistency and replicability of the framework developed. MCDA may help to structure discussions for heterogeneous treatment requests, providing predictability and robustness in decision making involving sensitive and complex situations.
Keywords: MCDA; assessment; drug policy; early access; multicriteria decision analysis; pharmaceuticals.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Directive 2001/83/EC of the European Parliament and of the Council of 6 November 2001 on the Community Code Relating to Medicinal Products for Human Use. Springer; Berlin/Heidelberg, Germany: 2001. European Parliament. - DOI
-
- World Health Organization—WHO . Medicines Reimbursement Policies in Europe. Regional Office for Europe; Geneva, Switzerland: 2018.

