Brain Iron Deficiency Changes the Stoichiometry of Adenosine Receptor Subtypes in Cortico-Striatal Terminals: Implications for Restless Legs Syndrome
- PMID: 35268590
- PMCID: PMC8911604
- DOI: 10.3390/molecules27051489
Brain Iron Deficiency Changes the Stoichiometry of Adenosine Receptor Subtypes in Cortico-Striatal Terminals: Implications for Restless Legs Syndrome
Abstract
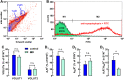
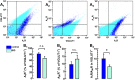
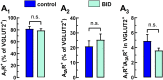
Brain iron deficiency (BID) constitutes a primary pathophysiological mechanism in restless legs syndrome (RLS). BID in rodents has been widely used as an animal model of RLS, since it recapitulates key neurochemical changes reported in RLS patients and shows an RLS-like behavioral phenotype. Previous studies with the BID-rodent model of RLS demonstrated increased sensitivity of cortical pyramidal cells to release glutamate from their striatal nerve terminals driving striatal circuits, a correlative finding of the cortical motor hyperexcitability of RLS patients. It was also found that BID in rodents leads to changes in the adenosinergic system, a downregulation of the inhibitory adenosine A1 receptors (A1Rs) and upregulation of the excitatory adenosine A2A receptors (A2ARs). It was then hypothesized, but not proven, that the BID-induced increased sensitivity of cortico-striatal glutamatergic terminals could be induced by a change in A1R/A2AR stoichiometry in favor of A2ARs. Here, we used a newly developed FACS-based synaptometric analysis to compare the relative abundance on A1Rs and A2ARs in cortico-striatal and thalamo-striatal glutamatergic terminals (labeled with vesicular glutamate transporters VGLUT1 and VGLUT2, respectively) of control and BID rats. It could be demonstrated that BID (determined by measuring transferrin receptor density in the brain) is associated with a selective decrease in the A1R/A2AR ratio in VGLUT1 positive-striatal terminals.
Keywords: adenosine A1 receptor; adenosine A2A receptor; brain iron deficiency; cortico-striatal terminals; restless legs syndrome; striatum; thalamo-striatal terminals.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

Similar articles
-
Adenosine receptors as markers of brain iron deficiency: Implications for Restless Legs Syndrome.Neuropharmacology. 2016 Dec;111:160-168. doi: 10.1016/j.neuropharm.2016.09.002. Epub 2016 Sep 4. Neuropharmacology. 2016. PMID: 27600688 Free PMC article.
-
Adenosine mechanisms and hypersensitive corticostriatal terminals in restless legs syndrome. Rationale for the use of inhibitors of adenosine transport.Adv Pharmacol. 2019;84:3-19. doi: 10.1016/bs.apha.2018.12.005. Epub 2019 Jan 18. Adv Pharmacol. 2019. PMID: 31229176 Free PMC article.
-
Pivotal Role of Adenosine Neurotransmission in Restless Legs Syndrome.Front Neurosci. 2018 Jan 8;11:722. doi: 10.3389/fnins.2017.00722. eCollection 2017. Front Neurosci. 2018. PMID: 29358902 Free PMC article.
-
Presynaptic adenosine receptor heteromers as key modulators of glutamatergic and dopaminergic neurotransmission in the striatum.Neuropharmacology. 2023 Feb 1;223:109329. doi: 10.1016/j.neuropharm.2022.109329. Epub 2022 Nov 11. Neuropharmacology. 2023. PMID: 36375695 Review.
-
New Insights into the Neurobiology of Restless Legs Syndrome.Neuroscientist. 2019 Apr;25(2):113-125. doi: 10.1177/1073858418791763. Epub 2018 Jul 26. Neuroscientist. 2019. PMID: 30047288 Free PMC article. Review.
Cited by
-
Review of the role of the endogenous opioid and melanocortin systems in the restless legs syndrome.Brain. 2024 Jan 4;147(1):26-38. doi: 10.1093/brain/awad283. Brain. 2024. PMID: 37633259 Free PMC article.
-
The ADORA1 mutation linked to early-onset Parkinson's disease alters adenosine A1-A2A receptor heteromer formation and function.Biomed Pharmacother. 2022 Dec;156:113896. doi: 10.1016/j.biopha.2022.113896. Epub 2022 Oct 21. Biomed Pharmacother. 2022. PMID: 36279718 Free PMC article.
-
Functional and pharmacological role of the dopamine D4 receptor and its polymorphic variants.Front Endocrinol (Lausanne). 2022 Sep 30;13:1014678. doi: 10.3389/fendo.2022.1014678. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36267569 Free PMC article. Review.
-
Lateral septum adenosine A2A receptors control stress-induced depressive-like behaviors via signaling to the hypothalamus and habenula.Nat Commun. 2023 Apr 5;14(1):1880. doi: 10.1038/s41467-023-37601-x. Nat Commun. 2023. PMID: 37019936 Free PMC article.
-
Circadian rhythm in restless legs syndrome.Front Neurol. 2023 Feb 23;14:1105463. doi: 10.3389/fneur.2023.1105463. eCollection 2023. Front Neurol. 2023. PMID: 36908590 Free PMC article. Review.
References
-
- Allen R.P., Picchietti D.L., Garcia-Borreguero D., Ondo W.G., Walters A.S., Winkelman J.W., Zucconi M., Ferri R., Trenkwalder C., Lee H.B. Restless Legs Syndrome Study Group. Restless legs syndrome/Willis–Ekbom disease diagnostic criteria: Updated International Restless Legs Syndrome Study Group (IRLSSG) consensus criteria—History, rationale, description, and significance. Sleep Med. 2014;15:860–873. doi: 10.1016/j.sleep.2014.03.025. - DOI - PubMed
MeSH terms
Substances
Grants and funding
- UIDB/04539/2020 and UIDP/04539/2020 of FCT/European Regional Development Fund (ERDF), through the Centro 2020 Regional Operational Program, and through the COMPETE 2020 - Operational Program for Competitiveness and Internationalization and Portuguese national funds
- Intramural Funds/DA/NIDA NIH HHS/United States
- LCF/PR/HP17/52190001/La Caixa Foundation

