Use of Gas Chromatography-Mass Spectrometry Techniques (GC-MS, GC-MS/MS and GC-QTOF) for the Characterization of Photooxidation and Autoxidation Products of Lipids of Autotrophic Organisms in Environmental Samples
- PMID: 35268730
- PMCID: PMC8911584
- DOI: 10.3390/molecules27051629
Use of Gas Chromatography-Mass Spectrometry Techniques (GC-MS, GC-MS/MS and GC-QTOF) for the Characterization of Photooxidation and Autoxidation Products of Lipids of Autotrophic Organisms in Environmental Samples
Abstract
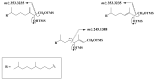
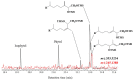
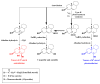
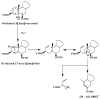
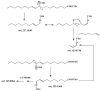
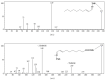
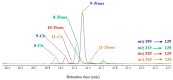
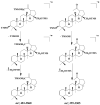
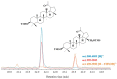
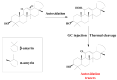
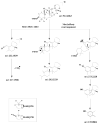
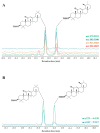
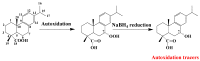
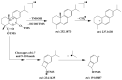
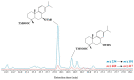
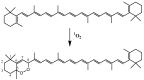
This paper reviews applications of gas chromatography-mass spectrometry techniques for the characterization of photooxidation and autoxidation products of lipids of senescent phototrophic organisms. Particular attention is given to: (i) the selection of oxidation products that are sufficiently stable under environmental conditions and specific to each lipid class and degradation route; (ii) the description of electron ionization mass fragmentation of trimethylsilyl derivatives of these compounds; and (iii) the use of specific fragment ions for monitoring the oxidation of the main unsaturated lipid components of phototrophs. The techniques best geared for this task were gas chromatography-quadrupole-time of flight to monitor fragment ions with very high resolution and accuracy, and gas chromatography-tandem mass spectrometry to monitor very selective transitions in multiple reaction monitoring mode. The extent of the degradation processes can only be estimated if the oxidation products are unaffected by fast secondary oxidation reactions, as it is notably the case of ∆5-sterols, monounsaturated fatty acids, chlorophyll phytyl side-chain, and di- and triterpenoids. In contrast, the primary degradation products of highly branched isoprenoid alkenes possessing more than one trisubstituted double bond, alkenones, carotenoids and polyunsaturated fatty acids, appear to be too unstable with respect to secondary oxidation or other reactions to serve for quantification in environmental samples.
Keywords: EI fragmentation; TMS derivatives; autoxidation; environment; gas chromatography-mass spectrometry; photooxidation; senescent phototrophs; specific tracers; unsaturated lipids.
Conflict of interest statement
The author declares no conflict of interest.
Figures





References
-
- Harwood J.L., Russell N.J. Lipids in Plants and Microbes. Springer; Dordrecht, The Netherlands: 1984. pp. 7–32.
-
- Ben-Amotz A., Tornabene T.G., Thomas W.H. Chemical profile of selected species of microalgae with emphasis on lipids. J. Phycol. 1985;21:72–81. doi: 10.1111/j.0022-3646.1985.00072.x. - DOI
-
- Volkman J.K. Lipid markers for marine organic matter. Handb. Environ. Chem. 2006;2:27–70.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

