Pyk2 Regulates MAMs and Mitochondrial Dynamics in Hippocampal Neurons
- PMID: 35269464
- PMCID: PMC8909471
- DOI: 10.3390/cells11050842
Pyk2 Regulates MAMs and Mitochondrial Dynamics in Hippocampal Neurons
Abstract
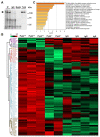
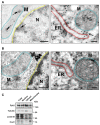
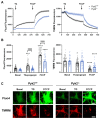
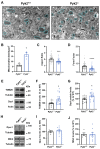
Pyk2 is a non-receptor tyrosine kinase enriched in hippocampal neurons, which can be activated by calcium-dependent mechanisms. In neurons, Pyk2 is mostly localised in the cytosol and dendritic shafts but can translocate to spines and/or to the nucleus. Here, we explore the function of a new localisation of Pyk2 in mitochondria-associated membranes (MAMs), a subdomain of ER-mitochondria surface that acts as a signalling hub in calcium regulation. To test the role of Pyk2 in MAMs' calcium transport, we used full Pyk2 knockout mice (Pyk2-/-) for in vivo and in vitro studies. Here we report that Pyk2-/- hippocampal neurons present increased ER-mitochondrial contacts along with defective calcium homeostasis. We also show how the absence of Pyk2 modulates mitochondrial dynamics and morphology. Taken all together, our results point out that Pyk2 could be highly relevant in the modulation of ER-mitochondria calcium efflux, affecting in turn mitochondrial function.
Keywords: ER-mitochondria contact sites; calcium; hippocampus; neuron.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Giralt A., Brito V., Chevy Q., Simonnet C., Otsu Y., Cifuentes-Díaz C., De Pins B., Coura R., Alberch J., Ginés S., et al. Pyk2 modulates hippocampal excitatory synapses and contributes to cognitive deficits in a Huntington’s disease model. Nat. Commun. 2017;8:15592. doi: 10.1038/ncomms15592. - DOI - PMC - PubMed
-
- Corvol J.-C., Valjent E., Toutant M., Enslen H., Irinopoulou T., Lev S., Hervé D., Girault J.-A. Depolarization Activates ERK and Proline-rich Tyrosine Kinase 2 (PYK2) Independently in Different Cellular Compartments in Hippocampal Slices. J. Biol. Chem. 2005;280:660–668. doi: 10.1074/jbc.M411312200. - DOI - PubMed
-
- Menegon A., Burgaya F., Baudot P., Dunlap D.D., Girault J.-A., Valtorta F. FAK + and PYK2/CAKβ, two related tyrosine kinases highly expressed in the central nervous system: Similarities and differences in the expression pattern. Eur. J. Neurosci. 1999;11:3777–3788. doi: 10.1046/j.1460-9568.1999.00798.x. - DOI - PubMed
-
- Faure C., Corvol J.-C., Toutant M., Valjent E., Hvalby O., Jensen V., El Messari S., Corsi J.-M., Kadare G., Girault J.-A. Calcineurin is essential for depolarization-induced nuclear translocation and tyrosine phosphorylation of PYK2 in neurons. J. Cell Sci. 2007;120:3034–3044. doi: 10.1242/jcs.009613. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

