Gut microbiota-derived bile acids in intestinal immunity, inflammation, and tumorigenesis
- PMID: 35271802
- PMCID: PMC8923532
- DOI: 10.1016/j.chom.2022.02.004
Gut microbiota-derived bile acids in intestinal immunity, inflammation, and tumorigenesis
Abstract
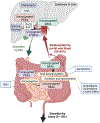
Inflammatory bowel disease (IBD) and colorectal cancer (CRC) are heterogeneous intestinal diseases that threaten the health of an increasing number of individuals as their lifestyles become westernized. New insights have been discovered with the development of various omics techniques, revealing that gut-microbiota-derived metabolites play important roles in maintaining intestinal homeostasis and modulating the progression of intestinal diseases from both metabolic and immunological perspectives. Clinical metagenomic and metabolomic studies have revealed links between microbial bile acid (BA) metabolism and IBD and CRC progression. Several BA-derived metabolites were recently been demonstrated to play a role in intestinal immunity, providing fresh insights into how BAs affect the course of IBD and CRC. In this review, we discuss recent studies on the involvement of gut microbiota-derived BAs in intestinal immunity, inflammation, and tumorigenesis along with human omics data to provide prospective insights into future prevention and treatment of IBD and CRC.
Keywords: bile acids; colorectal cancer; gut microbiota; inflammatory bowel diseases; omics.
Copyright © 2022. Published by Elsevier Inc.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures


References
-
- Axelson M, Ellis E, Mork B, Garmark K, Abrahamsson A, Bjorkhem I, Ericzon BG & Einarsson C (2000). Bile acid synthesis in cultured human hepatocytes: support for an alternative biosynthetic pathway to cholic acid. Hepatology, 31, 1305–1312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

