Metabolomic Impact of Lidocaine on a Triple Negative Breast Cancer Cell Line
- PMID: 35273500
- PMCID: PMC8902240
- DOI: 10.3389/fphar.2022.821779
Metabolomic Impact of Lidocaine on a Triple Negative Breast Cancer Cell Line
Abstract
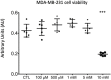
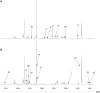
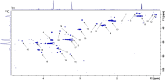
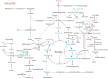
Background: Metabolomics and onco-anesthesia are two emerging research fields in oncology. Metabolomics (metabolites analysis) is a new diagnostic and prognostic tool that can also be used for predicting the therapeutic or toxic responses to anticancer treatments. Onco-anesthesia studies assess the impact of anesthesia on disease-free and overall survival after cancer surgery. It has been shown that local anesthetics (LA), particularly lidocaine (LIDO), exert antitumor properties both in vitro and in vivo and may alter the biologic fingerprints of cancer cells. As LA are known to impair mitochondrial bioenergetics and byproducts, the aim of the present study was to assess the impact of LIDO on metabolomic profile of a breast cancer cell line. Methods: Breast cancer MDA-MB-231 cells were exposed for 4 h to 0.5 mM LIDO or vehicle (n = 4). The metabolomic fingerprint was characterized by high resolution magic angle spinning NMR spectroscopy (HRMAS). The multivariate technique using the Algorithm to Determine Expected Metabolite Level Alteration (ADEMA) (Cicek et al., PLoS Comput. Biol., 2013, 9, e1002859), based on mutual information to identify expected metabolite level changes with respect to a specific condition, was used to determine the metabolites variations caused by LIDO. Results: LIDO modulates cell metabolites levels. Several pathways, including glutaminolysis, choline, phosphocholine and total choline syntheses were significantly downregulated in the LIDO group. Discussion: This is the first study assessing the impact of LIDO on metabolomic fingerprint of breast cancer cells. Among pathways downregulated by LIDO, many metabolites are reported to be associated with adverse prognosis when present at a high titer in breast cancer patients. These results fit with the antitumor properties of LIDO and suggest its impact on metabolomics profile of cancer cells. These effects of LIDO are of clinical significance because it is widely used for local anesthesia with cutaneous infiltration during percutaneous tumor biopsy. Future in vitro and preclinical studies are necessary to assess whether metabolomics analysis requires modification of local anesthetic techniques during tumor biopsy.
Keywords: anesthesia; cancer progression; cancer surgery; lidocaine; metabolomics; onco-anesthesia; perioperative period.
Copyright © 2022 Chamaraux-Tran, Muller, Pottecher, Diemunsch, Tomasetto, Namer and Dali-Youcef.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Lido-OH, a Hydroxyl Derivative of Lidocaine, Produced a Similar Local Anesthesia Profile as Lidocaine With Reduced Systemic Toxicities.Front Pharmacol. 2021 Sep 16;12:678437. doi: 10.3389/fphar.2021.678437. eCollection 2021. Front Pharmacol. 2021. PMID: 34603015 Free PMC article.
-
Buffered lidocaine 1%/epinephrine 1:100,000 with sodium bicarbonate (sodium hydrogen carbonate) in a 3:1 ratio is less painful than a 9:1 ratio: A double-blind, randomized, placebo-controlled, crossover trial.J Am Acad Dermatol. 2020 Jul;83(1):159-165. doi: 10.1016/j.jaad.2019.09.088. Epub 2020 Jan 17. J Am Acad Dermatol. 2020. PMID: 31958526 Clinical Trial.
-
[Cutaneous blood flow following subcutaneous infiltration of lidocaine with and without the addition of adrenaline or ornipressin].Reg Anaesth. 1990 Jun;13(4):97-100. Reg Anaesth. 1990. PMID: 2374837 German.
-
Evaluation of Cancer Metabolomics Using ex vivo High Resolution Magic Angle Spinning (HRMAS) Magnetic Resonance Spectroscopy (MRS).Metabolites. 2016 Mar 22;6(1):11. doi: 10.3390/metabo6010011. Metabolites. 2016. PMID: 27011205 Free PMC article. Review.
-
The Amide Local Anesthetic Lidocaine in Cancer Surgery-Potential Antimetastatic Effects and Preservation of Immune Cell Function? A Narrative Review.Front Med (Lausanne). 2017 Dec 20;4:235. doi: 10.3389/fmed.2017.00235. eCollection 2017. Front Med (Lausanne). 2017. PMID: 29326939 Free PMC article. Review.
Cited by
-
Trial watch: local anesthetics in cancer therapy.Oncoimmunology. 2024 Mar 17;13(1):2308940. doi: 10.1080/2162402X.2024.2308940. eCollection 2024. Oncoimmunology. 2024. PMID: 38504848 Free PMC article.
-
Anesthesia-related intervention for long-term survival and cancer recurrence following breast cancer surgery: A systematic review of prospective studies.PLoS One. 2023 Dec 21;18(12):e0296158. doi: 10.1371/journal.pone.0296158. eCollection 2023. PLoS One. 2023. PMID: 38127958 Free PMC article.
-
Comparative Metabolomics Study of the Impact of Articaine and Lidocaine on the Metabolism of SH-SY5Y Neuronal Cells.Metabolites. 2022 Jun 23;12(7):581. doi: 10.3390/metabo12070581. Metabolites. 2022. PMID: 35888705 Free PMC article.
-
Targeted Delivery of Lidocaine in Breast Cancer Cells via Zeolitic Imidazolate Framework-8 Nanoparticles.Chemphyschem. 2025 Aug 23;26(16):e202401128. doi: 10.1002/cphc.202401128. Epub 2025 Jun 24. Chemphyschem. 2025. PMID: 40556462 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

