The prognostic effect of residual tumor for advanced epithelial ovarian cancer treated with neoadjuvant chemotherapy or primary debulking surgery
- PMID: 35274489
- PMCID: PMC9302261
- DOI: 10.1002/cam4.4642
The prognostic effect of residual tumor for advanced epithelial ovarian cancer treated with neoadjuvant chemotherapy or primary debulking surgery
Abstract
Purpose: The role of neoadjuvant chemotherapy (NACT) and primary debulking surgery (PDS) in advanced epithelial ovarian cancer (EOC) remains controversial. This study aimed to investigate the prognosis between NACT and PDS in advanced EOC. We also investigated the prognostic effect of the residual tumor (RT) after NACT and PDS.
Methods: Patients with stage III-IV EOC diagnosed between 2010 and 2017 were included from the Surveillance, Epidemiology, and End Results (SEER) database. Chi-square test, multivariate logistic regression analysis, Kaplan-Meier curves, and Cox proportional hazards model were used for statistical analyses.
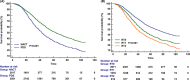
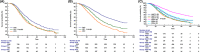
Results: A total of 5522 women patients were identified, 2017 (36.5%) and 3505 (63.5%) patients received NACT and PDS, respectively. There were 2971 (53.8%), 1637 (29.6%), and 914 (16.6%) patients who had no residual tumor, RT ≤1 cm, and RT >1 cm, respectively. There were 25.5% of patients receiving NACT in 2010 and 48.4% in 2017 (p < 0.001). Women treated with NACT were not related to a higher chance of complete resection than the PDS group (p = 0.098). Patients receiving PDS had significantly better cancer-specific survival (CSS) than those receiving NACT (p < 0.001). The 5-year CSS was 35.3% and 51.1% in those receiving NACT and PDS, respectively. In patients receiving NACT, those who had no residual tumor had significantly better CSS compared to those who had RT ≤1 cm (p < 0.001), while comparable CSS was found between those who had RT ≤1 cm and RT >1 cm (p = 0.442). In those receiving PDS, the CSS was decreased with a RT increase (p < 0.001).
Conclusions: Our study suggests that PDS may be the optimal procedure for the majority of advanced EOC patients. Complete resection of all residual diseases should be the goal with the increased utilization of NACT.
Keywords: debulking surgery; epithelial ovarian cancer; neoadjuvant chemotherapy; residual tumor; survival.
© 2022 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures


References
-
- Colombo N, Sessa C, du Bois A, et al. ESMO‐ESGO consensus conference recommendations on ovarian cancer: pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann Oncol. 2019;30:672‐705. - PubMed
-
- Armstrong DK, Alvarez RD, Bakkum‐Gamez JN, et al. Ovarian cancer, version 2.2020, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2021;19:191‐226. - PubMed
-
- Vergote I, Tropé CG, Amant F, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363:943‐953. - PubMed
-
- Kehoe S, Hook J, Nankivell M, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open‐label, randomised, controlled, non‐inferiority trial. Lancet. 2015;386:249‐257. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

