Complementary Medicine Use Amongst Patients with Metastatic Cancer Enrolled in Phase III Clinical Trials
- PMID: 35274718
- PMCID: PMC8914495
- DOI: 10.1093/oncolo/oyac020
Complementary Medicine Use Amongst Patients with Metastatic Cancer Enrolled in Phase III Clinical Trials
Abstract
Background: Complementary medicines (CM) are frequently used by patients with cancer. Controversy exists over the effectiveness and risk that CM may add to conventional cancer therapy. The incidence of CM use among patients enrolled in phase III clinical trials is unknown.
Methods: Medication lists from 6 international phase III clinical trials were retrospectively reviewed to identify patients using CM. Patients had metastatic breast, colorectal, or lung cancers. Quality of life, adverse events, overall survival, and progression-free survival were compared between CM users and non-users. Baseline differences between groups were adjusted with propensity score matching groups.
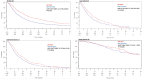
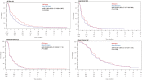
Results: Seven hundred and six of 3446 patients (20.5%) used at least one CM. CM use was highest among patients with breast cancer (35.6%). CM users had more favorable baseline prognostic factors (ECOG 0-1, non-smoking status, younger age, and fewer metastases). CM use was associated with lower rates of adverse events (50% vs. 62%, P = .002) and quality of life was similar between both groups. After adjustment with propensity score matching, CM use was also associated with longer overall survival in patients with lung cancer (adjusted hazard ratio 0.80, 95%CI, 0.68-0.94, P =.0054). However, several key control variables like EGFR status were not available.
Conclusion: One in 5 patients in phase III clinical trials report using CM. CM was not associated with worse cancer-specific outcomes. However, CM users had more favorable baseline prognostic factors, and likely other confounders that may have contributed to improved outcomes observed in the lung cohort. Physicians should monitor for CM use and potential interactions with clinical trial drugs.
Keywords: cancer; clinical trial; complementary medicine.
© The Author(s) 2022. Published by Oxford University Press.
Figures
References
-
- NCCIH. Complementary, Alternative, or Integrative Health: What’s In a Name?. https://nccih.nih.gov/health/integrative-health. Accessed October 23, 2019.
-
- Richardson MA, Sanders T, Palmer JL, Greisinger A, Singletary SE. Complementary/alternative medicine use in a comprehensive cancer center and the implications for oncology. J Clin Oncol. 2000;18(13):2505-2514. - PubMed
-
- Dy GK, Bekele L, Hanson LJ, et al. Complementary and alternative medicine use by patients enrolled onto phase I clinical trials. J Clin Oncol. 2004;22(23):4810-4815. - PubMed
-
- Naing A, Stephen SK, Frenkel M, et al. Prevalence of complementary medicine use in a phase 1 clinical trials program: the MD Anderson Cancer Center Experience. Cancer. 2011;117(22):5142-5150. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

