A novel method for estimating connectivity-based parcellation of the human brain from diffusion MRI: Application to an aging cohort
- PMID: 35274787
- PMCID: PMC9057102
- DOI: 10.1002/hbm.25773
A novel method for estimating connectivity-based parcellation of the human brain from diffusion MRI: Application to an aging cohort
Abstract
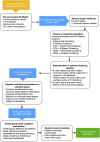
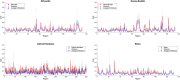
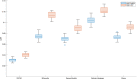
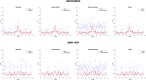
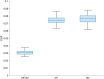
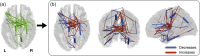
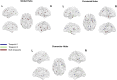
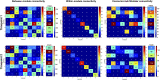
Connectivity-based parcellation (CBP) methods are used to define homogenous and biologically meaningful parcels or nodes-the foundations of brain network fingerprinting-by grouping voxels with similar patterns of brain connectivity. However, we still lack a gold standard method and the use of CBPs to study the aging brain remains scarce. Our study proposes a novel CBP method from diffusion MRI data and shows its potential to produce a more accurate characterization of the longitudinal alterations in brain network topology occurring in aging. For this, we constructed whole-brain connectivity maps from diffusion MRI data of two datasets: an aging cohort evaluated at two timepoints (mean interval time: 52.8 ± 7.24 months) and a normative adult cohort-MGH-HCP. State-of-the-art clustering techniques were used to identify the best performing technique. Furthermore, we developed a new metric (connectivity homogeneity fingerprint [CHF]) to evaluate the success of the final CBP in improving regional/global structural connectivity homogeneity. Our results show that our method successfully generates highly homogeneous parcels, as described by the significantly larger CHF score of the resulting parcellation, when compared to the original. Additionally, we demonstrated that the developed parcellation provides a robust anatomical framework to assess longitudinal changes in the aging brain. Our results reveal that aging is characterized by a reorganization of the brain's structural network involving the decrease of intra-hemispheric, increase of inter-hemispheric connectivity, and topological rearrangement. Overall, this study proposes a new methodology to perform accurate and robust evaluations of CBP of the human brain.
Keywords: aging; brain parcellation; clustering; diffusion MRI; network neuroscience; structural connectivity.
© 2022 The Authors. Human Brain Mapping published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare that they have no conflict of interests.
Figures


Similar articles
-
Reorganization of brain structural networks in aging: A longitudinal study.J Neurosci Res. 2021 May;99(5):1354-1376. doi: 10.1002/jnr.24795. Epub 2021 Feb 2. J Neurosci Res. 2021. PMID: 33527512 Free PMC article.
-
Individualized Cortical Parcellation Based on Diffusion MRI Tractography.Cereb Cortex. 2020 May 14;30(5):3198-3208. doi: 10.1093/cercor/bhz303. Cereb Cortex. 2020. PMID: 31814022
-
Constructing fMRI connectivity networks: a whole brain functional parcellation method for node definition.J Neurosci Methods. 2014 May 15;228:86-99. doi: 10.1016/j.jneumeth.2014.03.004. Epub 2014 Mar 25. J Neurosci Methods. 2014. PMID: 24675050
-
Connectivity-based parcellation: Critique and implications.Hum Brain Mapp. 2015 Dec;36(12):4771-92. doi: 10.1002/hbm.22933. Epub 2015 Sep 27. Hum Brain Mapp. 2015. PMID: 26409749 Free PMC article. Review.
-
Functional parcellation using time courses of instantaneous connectivity.Neuroimage. 2018 Apr 15;170:31-40. doi: 10.1016/j.neuroimage.2017.07.027. Epub 2017 Jul 14. Neuroimage. 2018. PMID: 28716715 Review.
References
-
- Adolfsson, A. , Ackerman, M. , & Brownstein, N. C. (2019). To cluster, or not to cluster: An analysis of clusterability methods. Pattern Recognition, 88, 13–26. 10.1016/j.patcog.2018.10.026 - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

