Multimethod quantitative benefit-risk assessment of treatments for moderate-to-severe osteoarthritis
- PMID: 35277997
- PMCID: PMC9543715
- DOI: 10.1111/bcp.15309
Multimethod quantitative benefit-risk assessment of treatments for moderate-to-severe osteoarthritis
Abstract
Objective: Demonstrate how benefit-risk profiles of systemic treatments for moderate-to-severe osteoarthritis (OA) can be compared using a quantitative approach accounting for patient preference.
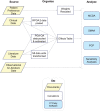
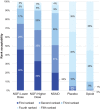
Study design and setting: This study used a multimethod benefit-risk modelling approach to quantifiably compare treatments of moderate-to-severe OA. In total four treatments and placebo were compared. Comparisons were based on four attributes identified as most important to patients. Patient Global Assessment of Osteoarthritis was included as a favourable effect. Unfavourable effects, or risks, included opioid dependence, nonfatal myocardial infarction and rapidly progressive OA leading to total joint replacement. Clinical data from randomized clinical trials, a meta-analysis of opioid dependence and a long-term study of celecoxib were mapped into value functions and weighted with patient preferences from a discrete choice experiment.
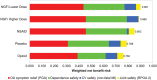
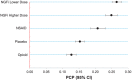
Results: Lower-dose NGFi had the highest weighted net benefit-risk score (0.901), followed by higher-dose NGFi (0.889) and NSAIDs (0.852), and the lowest score was for opioids (0.762). Lower-dose NGFi was the highest-ranked treatment option even when assuming a low incidence (0.34% instead of 4.7%) of opioid dependence (ie, opioid benefit-risk score 808) and accounting for both the uncertainty in clinical effect estimates (first rank probability 46% vs 20% for NSAIDs) and imprecision in patient preference estimates (predicted choice probability 0.26, 95% confidence interval [CI] 0.25-0.28 vs 0.21, 95% CI 0.19-0.23 for NSAIDs).
Conclusion: The multimethod approach to quantitative benefit-risk modelling allowed the interpretation of clinical data from the patient perspective while accounting for uncertainties in the clinical effect estimates and imprecision in patient preferences.
Keywords: benefit-risk assessment; nerve growth factor inhibitor; opioid; osteoarthritis; patient preference.
© 2022 Pfizer Inc. British Journal of Clinical Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
Figures
Similar articles
-
Patient preferences for osteoarthritis pain and chronic low back pain treatments in the United States: a discrete-choice experiment.Osteoarthritis Cartilage. 2020 Sep;28(9):1202-1213. doi: 10.1016/j.joca.2020.06.006. Epub 2020 Jul 8. Osteoarthritis Cartilage. 2020. PMID: 32652238
-
Intra-articular viscosupplementation with hylan g-f 20 to treat osteoarthritis of the knee: an evidence-based analysis.Ont Health Technol Assess Ser. 2005;5(10):1-66. Epub 2005 Jun 1. Ont Health Technol Assess Ser. 2005. PMID: 23074461 Free PMC article.
-
A discrete-choice experiment of United Kingdom patients' willingness to risk adverse events for improved function and pain control in osteoarthritis.Osteoarthritis Cartilage. 2013 Feb;21(2):289-97. doi: 10.1016/j.joca.2012.11.007. Epub 2012 Nov 24. Osteoarthritis Cartilage. 2013. PMID: 23182815
-
Tramadol for osteoarthritis.Cochrane Database Syst Rev. 2019 May 27;5(5):CD005522. doi: 10.1002/14651858.CD005522.pub3. Cochrane Database Syst Rev. 2019. PMID: 31132298 Free PMC article.
-
Outcome measures in placebo-controlled trials of osteoarthritis: responsiveness to treatment effects in the REPORT database.Osteoarthritis Cartilage. 2011 May;19(5):483-92. doi: 10.1016/j.joca.2011.02.020. Epub 2011 Mar 23. Osteoarthritis Cartilage. 2011. PMID: 21396467 Review.
Cited by
-
Evaluating the balance of benefits and harms in chronic pain clinical trials: prioritizing individual participants over individual outcomes.Reg Anesth Pain Med. 2024 May 7;49(5):363-367. doi: 10.1136/rapm-2023-104809. Reg Anesth Pain Med. 2024. PMID: 37963675 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

