Mitochondrial Dysfunction: An Emerging Link in the Pathophysiology of Cardiorenal Syndrome
- PMID: 35282359
- PMCID: PMC8914047
- DOI: 10.3389/fcvm.2022.837270
Mitochondrial Dysfunction: An Emerging Link in the Pathophysiology of Cardiorenal Syndrome
Abstract
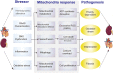
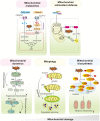
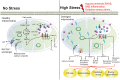
The crosstalk between the heart and kidney is carried out through various bidirectional pathways. Cardiorenal syndrome (CRS) is a pathological condition in which acute or chronic dysfunction in the heart or kidneys induces acute or chronic dysfunction of the other organ. Complex hemodynamic factors and biochemical and hormonal pathways contribute to the development of CRS. In addition to playing a critical role in generating metabolic energy in eukaryotic cells and serving as signaling hubs during several vital processes, mitochondria rapidly sense and respond to a wide range of stress stimuli in the external environment. Impaired adaptive responses ultimately lead to mitochondrial dysfunction, inducing cell death and tissue damage. Subsequently, these changes result in organ failure and trigger a vicious cycle. In vitro and animal studies have identified an important role of mitochondrial dysfunction in heart failure (HF) and chronic kidney disease (CKD). Maintaining mitochondrial homeostasis may be a promising therapeutic strategy to interrupt the vicious cycle between HF and acute kidney injury (AKI)/CKD. In this review, we hypothesize that mitochondrial dysfunction may also play a central role in the development and progression of CRS. We first focus on the role of mitochondrial dysfunction in the pathophysiology of HF and AKI/CKD, then discuss the current research evidence supporting that mitochondrial dysfunction is involved in various types of CRS.
Keywords: cardiorenal syndrome; heart failure; inflammation; kidney failure; mitochondrial dysfunction; oxidative stress.
Copyright © 2022 Shi, Zhang, Li, Xu, Lv, Jia, Chai, Xue, Li, Wang, Wu, Song and Hu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Mitochondrial Dysfunction in Cardiorenal Syndrome.Antioxid Redox Signal. 2016 Aug 1;25(4):200-7. doi: 10.1089/ars.2016.6654. Antioxid Redox Signal. 2016. PMID: 27349577 Review.
-
Update on the Classification and Pathophysiological Mechanisms of Pediatric Cardiorenal Syndromes.Children (Basel). 2021 Jun 22;8(7):528. doi: 10.3390/children8070528. Children (Basel). 2021. PMID: 34206173 Free PMC article. Review.
-
Target organ cross talk in cardiorenal syndrome: animal models.Am J Physiol Renal Physiol. 2012 Nov 1;303(9):F1253-63. doi: 10.1152/ajprenal.00392.2012. Epub 2012 Aug 22. Am J Physiol Renal Physiol. 2012. PMID: 22914779 Review.
-
Cardiorenal syndrome type 1: pathophysiological crosstalk leading to combined heart and kidney dysfunction in the setting of acutely decompensated heart failure.J Am Coll Cardiol. 2012 Sep 18;60(12):1031-42. doi: 10.1016/j.jacc.2012.01.077. Epub 2012 Jul 25. J Am Coll Cardiol. 2012. PMID: 22840531 Review.
-
The hemodynamic and nonhemodynamic crosstalk in cardiorenal syndrome type 1.Cardiorenal Med. 2014 Aug;4(2):103-12. doi: 10.1159/000362650. Epub 2014 May 14. Cardiorenal Med. 2014. PMID: 25254032 Free PMC article. Review.
Cited by
-
Omega-3 Fatty Acids Modify Drp1 Expression and Activate the PINK1-Dependent Mitophagy Pathway in the Kidney and Heart of Adenine-Induced Uremic Rats.Biomedicines. 2024 Sep 15;12(9):2107. doi: 10.3390/biomedicines12092107. Biomedicines. 2024. PMID: 39335620 Free PMC article.
-
Multiple roles of mitochondrial autophagy receptor FUNDC1 in mitochondrial events and kidney disease.Front Cell Dev Biol. 2024 Oct 9;12:1453365. doi: 10.3389/fcell.2024.1453365. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 39445333 Free PMC article. Review.
-
NAC Pre-Administration Prevents Cardiac Mitochondrial Bioenergetics, Dynamics, Biogenesis, and Redox Alteration in Folic Acid-AKI-Induced Cardio-Renal Syndrome Type 3.Antioxidants (Basel). 2023 Aug 10;12(8):1592. doi: 10.3390/antiox12081592. Antioxidants (Basel). 2023. PMID: 37627587 Free PMC article.
-
The Role of Oxidative Stress as a Mechanism in the Pathogenesis of Acute Heart Failure in Acute Kidney Injury.Diagnostics (Basel). 2024 Sep 23;14(18):2094. doi: 10.3390/diagnostics14182094. Diagnostics (Basel). 2024. PMID: 39335773 Free PMC article. Review.
-
Mitochondrial Dysfunction in the Cardio-Renal Axis.Int J Mol Sci. 2023 May 3;24(9):8209. doi: 10.3390/ijms24098209. Int J Mol Sci. 2023. PMID: 37175915 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

