Shikonin Attenuates Cochlear Spiral Ganglion Neuron Degeneration by Activating Nrf2-ARE Signaling Pathway
- PMID: 35283722
- PMCID: PMC8908960
- DOI: 10.3389/fnmol.2022.829642
Shikonin Attenuates Cochlear Spiral Ganglion Neuron Degeneration by Activating Nrf2-ARE Signaling Pathway
Abstract
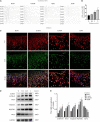
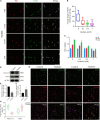
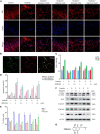
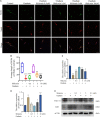
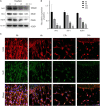
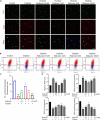
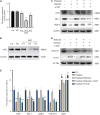
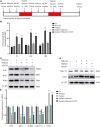
The molecular mechanisms that regulate the proliferation and differentiation of inner ear spiral ganglion cells (SGCs) remain largely unknown. Shikonin (a naphthoquinone pigment isolated from the traditional Chinese herbal medicine comfrey root) has anti-oxidation, anti-apoptosis and promoting proliferation and differentiation effects on neural progenitor cells. To study the protective effect of shikonin on auditory nerve damage, we isolated spiral ganglion neuron cells (SGNs) and spiral ganglion Schwann cells (SGSs) that provide nutrients in vitro and pretreated them with shikonin. We found that shikonin can reduce ouabain, a drug that can selectively destroy SGNs and induce auditory nerve damage, caused SGNs proliferation decreased, neurite outgrowth inhibition, cells apoptosis and mitochondrial depolarization. In addition, we found that shikonin can increase the expression of Nrf2 and its downstream molecules HO-1 and NQO1, thereby enhancing the antioxidant capacity of SGNs and SGSs, promoting cells proliferation, and inhibiting cells apoptosis by activating the Nrf2/antioxidant response elements (ARE) signal pathway. However, knockdown of Nrf2 rescued the protective effect of shikonin on SGNs and SGSs damage. In addition, we injected shikonin pretreatment into mouse that ouabain-induced hearing loss and found that shikonin pretreatment has a defensive effect on auditory nerve damage. In summary, the results of this study indicate that shikonin could attenuate the level of oxidative stress in SGNs and SGSs through the Nrf2-ARE signaling pathway activated, induce the proliferation and differentiation of SGNs, and thereby improve the neurological hearing damage in mice. Therefore, shikonin may be a candidate therapeutic drug for endogenous antioxidants that can be used to treat neurological deafness.
Keywords: Nrf2-ARE; auditory nerve damage; ouabain; shikonin; spiral ganglion cells.
Copyright © 2022 Du, Zhou, Shi, Xia, Wang, Guo, Hu, Zhang, Yang, Zhu, Teng, Liu and Zhao.
Conflict of interest statement
HD was employed by the company Qilu Pharmaceutical Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Albarakati A. J. A., Baty R. S., Aljoudi A. M., Habotta O. A., Elmahallawy E. K., Kassab R. B., et al. (2020). Luteolin protects against lead acetate-induced nephrotoxicity through antioxidant, anti-inflammatory, anti-apoptotic, and Nrf2/HO-1 signaling pathways. Mol. Biol. Rep. 47 2591–2603. 10.1007/s11033-020-05346-1 - DOI - PubMed
-
- Buchman C. A., Gifford R. H., Haynes D. S., Lenarz T., O’Donoghue G., Adunka O., et al. (2020). Unilateral cochlear implants for severe, profound, or moderate sloping to profound bilateral sensorineural hearing loss: a systematic review and consensus statements. JAMA Otolaryngol. Head Neck Surg. 146 942–953. 10.1001/jamaoto.2020.0998 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

