Dishonest Signaling in Microbial Conflicts
- PMID: 35283822
- PMCID: PMC8914469
- DOI: 10.3389/fmicb.2022.812763
Dishonest Signaling in Microbial Conflicts
Abstract
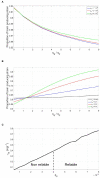
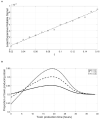
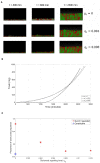
Quorum sensing is a cell-cell communication system that bacteria use to express social phenotypes, such as the production of extracellular enzymes or toxins, at high cell densities when these phenotypes are most beneficial. However, many bacterial strains are known to lack a sensing mechanism for quorum signals, despite having the gene responsible for releasing the signals to the environment. The aim of this article is 2-fold. First, we utilize mathematical modeling and signaling theory to elucidate the advantage that a bacterial species can gain by releasing quorum signals, while not being able to sense them, in the context of ecological competition with a focal quorum sensing species, by reducing the focal species' ability to optimize the timing of expression of the quorum sensing regulated phenotype. Additionally, the consequences of such "dishonest signaling," signaling that has evolved to harm the signal's receiver, on the focal quorum sensing species are investigated. It is found that quorum sensing bacteria would have to incur an additional, strategic, signaling cost in order to not suffer a reduction in fitness against dishonest signaling strains. Also, the concept of the Least Expensive Reliable Signal is introduced and applied to study how the properties of the regulated phenotype affect the metabolic investment in signaling needed by the quorum sensing bacteria to withstand dishonest signaling.
Keywords: bacterial social interactions; individual based modeling; mathematical modeling; quorum sensing; signaling theory.
Copyright © 2022 Hashem and Van Impe.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







References
-
- Bassler B. L. (1999). How bacteria talk to each other: regulation of gene expression by quorum sensing. Curr. Opin. Microbiol. 2, 582–587. - PubMed
-
- Bassler B. L., Wright M., Showalter R. E., Silverman M. R. (1993). Intercellular signalling in vibrio harveyi: sequence and function of genes regulating expression of luminescence. Mol Microbiol. 9, 773–786. - PubMed
LinkOut - more resources
Full Text Sources

