Gynecologic oncology HPV vaccination practice patterns: Investigating practice barriers, knowledge gaps and opportunities for maximizing cervical cancer prevention
- PMID: 35284612
- PMCID: PMC8907676
- DOI: 10.1016/j.gore.2022.100952
Gynecologic oncology HPV vaccination practice patterns: Investigating practice barriers, knowledge gaps and opportunities for maximizing cervical cancer prevention
Abstract
Objective: HPV vaccination is an important form of cancer prevention. Gynecologic oncologists have an opportunity to improve adult vaccination rates. We aimed to describe current HPV vaccination practices and barriers to vaccination reported by gynecologic oncologists.
Methods: An online survey was developed, pilot tested and sent to U.S. members of the Society of Gynecologic Oncology.
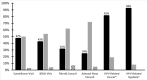
Results: Of the 226 respondents, most were female (73%), < 45 years old (64%) and practiced in urban (60%) and academic settings (69%). Ninety percent had recommended the HPV vaccine in the past year. Nearly half (47%) had facilitated vaccination by: administering the HPV vaccine in clinic (40%), stocking the vaccine (35%), or prescribing the vaccine (30%). Recommending the vaccine was associated with higher outpatient volume, practicing in the South vs. Northeast, and having higher levels of vaccine knowledge.Of the 90% who recommended the vaccine, 60% did not prescribe or know if they could prescribe the vaccine in their state. Prioritization of cancer treatment was the most commonly reported barrier to HPV vaccination (88%). Approximately half of providers reported other systems-level hinderances such as high cost of stocking the vaccine, clinic flow disruption, or uncertainty surrounding insurance coverage. Almost all recommenders offered the vaccine at HPV-related dysplasia (92%) or cancer (80%) visits, while only 24-50% offered it at non-HPV-related visits.
Conclusions: These survey results identify patient, provider, and systems-level barriers that could be targeted to help increase adult HPV vaccination in gynecologic oncology clinics.
Keywords: Barriers; Gynecologic oncologists; HPV vaccine; HPV vaccine champion; HPV-related Cancer prevention; Survey.
© 2022 The Authors. Published by Elsevier Inc.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

References
-
- Administration USFaD, 2018. FDA approves expanded use Gardasil 9 to include individuals 27 through 45 years old 2018. p. https://www.fda.gov/news-events/press-announcements/fda-approves-expande....
-
- Human Papillomavirus Vaccination ACOG Committee Opinion Summary, Number 809. Obstet. Gynecol. 2020;136(2):435–436. - PubMed
-
- Lichter K., Krause D., Xu J., Tsai S.H.L., Hage C., Weston E., Eke A., Levinson K. Adjuvant Human Papillomavirus Vaccine to Reduce Recurrent Cervical Dysplasia in Unvaccinated Women: A Systematic Review and Meta-analysis. Obstet. Gynecol. 2020;135(5):1070–1083. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
