Safety and immunogenicity of an inactivated recombinant Newcastle disease virus vaccine expressing SARS-CoV-2 spike: Interim results of a randomised, placebo-controlled, phase 1 trial
- PMID: 35284808
- PMCID: PMC8903824
- DOI: 10.1016/j.eclinm.2022.101323
Safety and immunogenicity of an inactivated recombinant Newcastle disease virus vaccine expressing SARS-CoV-2 spike: Interim results of a randomised, placebo-controlled, phase 1 trial
Abstract
Background: Production of affordable coronavirus disease 2019 (COVID-19) vaccines in low- and middle-income countries is needed. NDV-HXP-S is an inactivated egg-based recombinant Newcastle disease virus vaccine expressing the spike (S) protein of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). It's being developed by public sector manufacturers in Thailand, Vietnam, and Brazil; herein are initial results from Thailand.
Methods: This phase 1 stage of a randomised, dose-escalation, observer-blind, placebo-controlled, phase 1/2 trial was conducted at the Vaccine Trial Centre, Mahidol University (Bangkok). Healthy males and non-pregnant females, aged 18-59 years and negative for SARS-CoV-2 antibodies, were eligible. Participants were randomised to receive one of six treatments by intramuscular injection twice, 28 days apart: 1 µg, 1 µg+CpG1018 (a toll-like receptor 9 agonist), 3 µg, 3 µg+CpG1018, 10 µg, or placebo. Participants and personnel assessing outcomes were masked to treatment. The primary outcomes were solicited and spontaneously reported adverse events (AEs) during 7 and 28 days after each vaccination, respectively. Secondary outcomes were immunogenicity measures (anti-S IgG and pseudotyped virus neutralisation). An interim analysis assessed safety at day 57 in treatment-exposed individuals and immunogenicity through day 43 per protocol. ClinicalTrials.gov (NCT04764422).
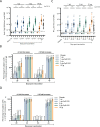
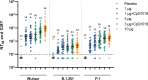
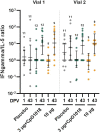
Findings: Between March 20 and April 23, 2021, 377 individuals were screened and 210 were enroled (35 per group); all received dose one; five missed dose two. The most common solicited AEs among vaccinees, all predominantly mild, were injection site pain (<63%), fatigue (<35%), headache (<32%), and myalgia (<32%). The proportion reporting a vaccine-related AE ranged from 5·7% to 17·1% among vaccine groups and was 2·9% in controls; there was no vaccine-related serious adverse event. The 10 µg formulation's immunogenicity ranked best, followed by 3 µg+CpG1018, 3 µg, 1 µg+CpG1018, and 1 µg formulations. On day 43, the geometric mean concentrations of 50% neutralising antibody ranged from 122·23 international units per mL (IU/mL; 1 µg, 95% confidence interval (CI) 86·40-172·91) to 474·35 IU/mL (10 µg, 95% CI 320·90-701·19), with 93·9% to 100% of vaccine groups attaining a ≥ 4-fold increase over baseline.
Interpretation: NDV-HXP-S had an acceptable safety profile and potent immunogenicity. The 3 µg and 3 µg+CpG1018 formulations advanced to phase 2.
Funding: National Vaccine Institute (Thailand), National Research Council (Thailand), Bill & Melinda Gates Foundation, National Institutes of Health (USA).
© 2022 The Author(s).
Conflict of interest statement
PN, SSur, SPra, SPuk, RK, RSin, NS, SThe, TV, KP, and PW are salaried employees of the Government Pharmaceutical Organization (Thailand). RH is a paid consultant to PATH. WS reports royalty payments from Avimex. AGS reports financial support from the U.S. NIAID (Centers of Excellence for Influenza Research and Response 75N93021C00014, Collaborative Influenza Vaccine Innovation Centers contract 75N93019C00051). The AGS laboratory has received research support from Pfizer, Senhwa Biosciences, Kenall Manufacturing, Avimex, Johnson & Johnson, Dynavax, 7Hills Pharma, Pharmamar, ImmunityBio, Accurius, Nanocomposix, Hexamer, N-fold LLC, Model Medicines, and Merck. AGS has consulting agreements for the following companies involving cash and/or stock: Vivaldi Biosciences, Contrafect, 7Hills Pharma, Avimex, Vaxalto, Pagoda, Accurius, Esperovax, Farmak, Applied Biological Laboratories, Curelab Oncology, Curelab Veterinary, and Pfizer; he also has received royalties (Merck, Astrazeneca, BI Vetmedica, Avimex, Regeneron), payment for lectures (Seqirus, Pharmamar), and participates in scientific advisory boards for New York State on Covid-19 vaccines, Accurius, Vaxalto, and Pfizer PP reports financial support from the U.S. NIAID (Centers of Excellence for Influenza Research and Response 75N93021C00014, P01 AI097092–07, R01 AI145870–03, Collaborative Influenza Vaccine Innovation Centers contract 75N93019C00051), payments involving cash and/or stock from Avimex, Vaxalto, and Accurius, royalty payments from Astrazeneca, BI Vetmedica, and Avimex; he also reports participation on the following advisory boards: New York State advisory board on Covid-19 vaccines, Accurius, and Vaxalto. PP also reports philanthropic grants or contracts through Mount Sinai. FK reports financial support from the U.S. NIAID (Collaborative Influenza Vaccine Innovation Centers contract 75N93019C00051, centre of Excellence for Influenza Research and Surveillance contract HHSN272201400008C), the JPB Foundation and the Open Philanthropy Project (research grant 2020–215,611, 5384), Pfizer, and the U.S. NCI ( contract 75N91019D00024, task order 75N91020F00003); he also has received royalties (Avimex), consulting fees (Pfizer, Seqirus, Third Rock Ventures, and Avimex), and payment for academic lectures during the past two years. NB reports participation as an AACR board member and support from SITC for travel and meeting attendance. CLH and JSM report financial support from the Bill & Melinda Gates Foundation and the U.S. NIH. The vaccine administered in this study was developed by faculty members at the Icahn School of Medicine at Mount Sinai including WS, PP, AGS, and FK. Mount Sinai has filed patent applications relating to SARS-CoV-2 serological assays and the NDV-based SARS-CoV-2 vaccine; the institution and its faculty inventors could benefit financially. JSM and CLH are inventors on a patent application concerning the Hexapro stabilized SARS-CoV-2 spike protein that was filed by the University of Texas at Austin and has been licensed to multiple entities; the university and its faculty inventors could benefit financially. All other authors have nothing to declare.
Figures

Update of
-
Safety and Immunogenicity of an Inactivated Recombinant Newcastle Disease Virus Vaccine Expressing SARS-CoV-2 Spike: Interim Results of a Randomised, Placebo-Controlled, Phase 1/2 Trial.medRxiv [Preprint]. 2021 Sep 22:2021.09.17.21263758. doi: 10.1101/2021.09.17.21263758. medRxiv. 2021. Update in: EClinicalMedicine. 2022 Mar 08;45:101323. doi: 10.1016/j.eclinm.2022.101323. PMID: 34580673 Free PMC article. Updated. Preprint.
References
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

