Fecal sphingolipids predict parenteral nutrition-associated cholestasis in the neonatal intensive care unit
- PMID: 35285019
- PMCID: PMC9468188
- DOI: 10.1002/jpen.2374
Fecal sphingolipids predict parenteral nutrition-associated cholestasis in the neonatal intensive care unit
Abstract
Background: Parenteral nutrition-associated cholestasis (PNAC) in the neonatal intensive care unit (NICU) causes significant morbidity and associated healthcare costs. Laboratory detection of PNAC currently relies on elevated serum conjugated bilirubin levels in the aftermath of impaired bile flow. Here, we sought to identify fecal biomarkers, which when integrated with clinical data, would better predict risk for developing PNAC.
Methods: Using untargeted metabolomics in 200 serial stool samples from 60 infants, we applied statistical and machine learning approaches to identify clinical features and metabolic biomarkers with the greatest associative potential for risk of developing PNAC. Stools were collected prospectively from infants receiving PN with soybean oil-based lipid emulsion at a level IV NICU.
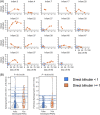
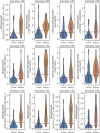
Results: Low birth weight, extreme prematurity, longer duration of PN, and greater number of antibiotic courses were all risk factors for PNAC (P < 0.05). We identified 78 stool biomarkers with early predictive potential (P < 0.05). From these 78 biomarkers, we further identified 12 sphingomyelin lipids with high association for the development of PNAC in precholestasis stool samples when combined with birth anthropometry.
Conclusion: We demonstrate the potential for stool metabolomics to enhance early identification of PNAC risk. Earlier detection of high-risk infants would empower proactive mitigation with alterations to PN for at-risk infants and optimization of energy nutrition with PN for infants at lower risk.
Keywords: early detection; infant; metabolomics; neonatal intensive care unit; parenteral nutrition-associated cholestasis; sphingomyelin; stool.
© 2022 The Authors. Journal of Parenteral and Enteral Nutrition published by Wiley Periodicals LLC on behalf of American Society for Parenteral and Enteral Nutrition.
Conflict of interest statement
None declared.
Figures



References
-
- Venigalla S, Gourley GR. Neonatal cholestasis. Semin Perinatol. 2004;28(5):348‐355. - PubMed
-
- Hsieh M‐H, Pai W, Tseng H‐I, Yang S‐N, Lu C‐C, Chen H‐L. Parenteral nutrition‐associated cholestasis in premature babies: risk factors and predictors. Pediatr Neonatol. 2009;50(5):202‐207. - PubMed
-
- Christensen RD, Henry E, Wiedmeier SE, Burnett J, Lambert DK. Identifying patients, on the first day of life, at high‐risk of developing parenteral nutrition‐associated liver disease. J Perinatol. 2007;27(5):284‐290. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

