Immunomodulation by endothelial cells - partnering up with the immune system?
- PMID: 35288707
- PMCID: PMC8920067
- DOI: 10.1038/s41577-022-00694-4
Immunomodulation by endothelial cells - partnering up with the immune system?
Abstract
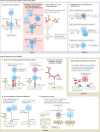
Blood vessel endothelial cells (ECs) have long been known to modulate inflammation by regulating immune cell trafficking, activation status and function. However, whether the heterogeneous EC populations in various tissues and organs differ in their immunomodulatory capacity has received insufficient attention, certainly with regard to considering them for alternative immunotherapy. Recent single-cell studies have identified specific EC subtypes that express gene signatures indicative of phagocytosis or scavenging, antigen presentation and immune cell recruitment. Here we discuss emerging evidence suggesting a tissue-specific and vessel type-specific immunomodulatory role for distinct subtypes of ECs, here collectively referred to as 'immunomodulatory ECs' (IMECs). We propose that IMECs have more important functions in immunity than previously recognized, and suggest that these might be considered as targets for new immunotherapeutic approaches.
© 2022. Springer Nature Limited.
Conflict of interest statement
P.C. declares associations with Montis Biosciences, Leuven, Belgium, of which he was a scientific co-founder. The remaining authors declare no competing interests.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

