Improvement of the antibody-dependent respiratory burst assay for assessing protective immune responses to malaria
- PMID: 35291880
- PMCID: PMC8924748
- DOI: 10.1098/rsob.210288
Improvement of the antibody-dependent respiratory burst assay for assessing protective immune responses to malaria
Abstract
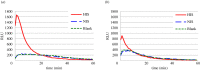
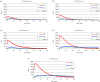
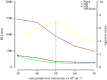
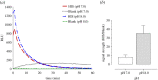
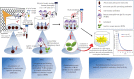
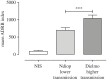
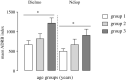
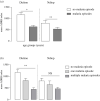
The antibody-dependent respiratory burst (ADRB) assay is a sensitive isoluminol-based chemiluminescence (CL) functional assay designed to assess the capacity of opsonizing antibodies against merozoites to induce neutrophil respiratory burst. ADRB was shown to measure protective immunity against malaria in endemic areas, but the assay needed further improvement to ensure better sensitivity and reproducibility. Here, we adjusted parameters such as the freezing-thawing procedure of merozoites, merozoites's concentration and the buffer solution's pH, and we used the improved assay to measure ADRB activity of 207 sera from 97 and 110 individuals living, respectively, in Dielmo and Ndiop villages with differing malaria endemicity. The improvement led to increased CL intensity and assay sensitivity, and a higher reproducibility. In both areas, ADRB activity correlated with malaria endemicity and individual's age discriminated groups with and without clinical malaria episodes, and significantly correlated with in vivo clinical protection from Plasmodium falciparum malaria. Our results demonstrate that the improved ADRB assay can be valuably used to assess acquired immunity during monitoring by control programmes and/or clinical trials.
Keywords: Plasmodium falciparum; functional assay; malaria; merozoites; neutrophils; respiratory burst.
Conflict of interest statement
We declare we have no competing interests.
Figures
References
-
- WHO. 2020. World malaria report 2019. Geneva, Switzerland: World Health Organization, 2019.
-
- Perraut R, Marrama L, Diouf B, Sokhna C, Tall A, Nabeth P, Trape JF, Longacre S, Mercereau-Puijalon O. 2005. Antibodies to the conserved C-terminal domain of the Plasmodium falciparum merozoite surface protein 1 and to the merozoite extract and their relationship with in vitro inhibitory antibodies and protection against clinical malaria in a Senegalese village. J. Infect. Dis. 191, 264-271. ( 10.1086/426398) - DOI - PubMed
-
- Nebie I, et al. 2008. Humoral responses to Plasmodium falciparum blood-stage antigens and association with incidence of clinical malaria in children living in an area of seasonal malaria transmission in Burkina Faso, West Africa. Infect. Immun. 76, 759-766. ( 10.1128/IAI.01147-07) - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
