Global update on the susceptibilities of human influenza viruses to neuraminidase inhibitors and the cap-dependent endonuclease inhibitor baloxavir, 2018-2020
- PMID: 35292289
- PMCID: PMC9254721
- DOI: 10.1016/j.antiviral.2022.105281
Global update on the susceptibilities of human influenza viruses to neuraminidase inhibitors and the cap-dependent endonuclease inhibitor baloxavir, 2018-2020
Abstract
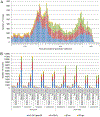
Global analysis of the susceptibility of influenza viruses to neuraminidase (NA) inhibitors (NAIs) and the polymerase acidic (PA) inhibitor (PAI) baloxavir was conducted by five World Health Organization Collaborating Centres for Reference and Research on Influenza during two periods (May 2018-May 2019 and May 2019-May 2020). Combined phenotypic and NA sequence-based analysis revealed that the global frequency of viruses displaying reduced or highly reduced inhibition (RI or HRI) or potential to show RI/HRI by NAIs remained low, 0.5% (165/35045) and 0.6% (159/26010) for the 2018-2019 and 2019-2020 periods, respectively. The most common amino acid substitution was NA-H275Y (N1 numbering) conferring HRI by oseltamivir and peramivir in A(H1N1)pdm09 viruses. Combined phenotypic and PA sequence-based analysis showed that the global frequency of viruses showing reduced susceptibility to baloxavir or carrying substitutions associated with reduced susceptibility was low, 0.5% (72/15906) and 0.1% (18/15692) for the 2018-2019 and 2019-2020 periods, respectively. Most (n = 61) of these viruses had I38→T/F/M/S/L/V PA amino acid substitutions. In Japan, where baloxavir use was highest, the rate was 4.5% (41/919) in the 2018-2019 period and most of the viruses (n = 32) had PA-I38T. Zoonotic viruses isolated from humans (n = 32) in different countries did not contain substitutions in NA associated with NAI RI/HRI phenotypes. One A(H5N6) virus had a dual substitution PA-I38V + PA-E199G, which may reduce susceptibility to baloxavir. Therefore, NAIs and baloxavir remain appropriate choices for the treatment of influenza virus infections, but close monitoring of antiviral susceptibility is warranted.
Keywords: Antiviral; Baloxavir; Influenza; Neuraminidase inhibitor; Polymerase inhibitor; Reduced susceptibility.
Copyright © 2022 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




References
-
- Air GM, Els MC, Brown LE, Laver WG, Webster RG, 1985. Location of antigenic sites on the three-dimensional structure of the influenza N2 virus neuraminidase. Virol 145 (2), 237–248. - PubMed
-
- Boltz DA, Douangngeun B, Phommachanh P, Sinthasak S, Mondry R, Obert C, Seiler P, Keating R, Suzuki Y, Hiramatsu H, Govorkova EA, Webster RG, 2010. Emergence of H5N1 avian influenza viruses with reduced sensitivity to neuraminidase inhibitors and novel reassortants in Lao People’s Democratic Republic. J. Gen. Virol 91 (4), 949–959. - PMC - PubMed
-
- Collins PJ, Haire LF, Lin YP, Liu J, Russell RJ, Walker PA, Martin SR, Daniels RS, Gregory V, Skehel JJ, Gamblin SJ, Hay AJ, 2009. Structural basis for oseltamivir resistance of influenza viruses. Vaccine 27 (45), 6317–6323. - PubMed
-
- Dharan NJ, Gubareva LV, Meyer JJ, Okomo-Adhiambo M, McClinton RC, Marshall SA, St George K, Epperson S, Brammer L, Klimov AI, Bresee JS, Fry AM, Oseltamivir-Resistance Working Group., 2009. Infections with oseltamivir-resistant influenza A(H1N1) virus in the United States. JAMA 301 (10), 1034–1041. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

