A clicking confinement strategy to fabricate transition metal single-atom sites for bifunctional oxygen electrocatalysis
- PMID: 35294235
- PMCID: PMC8926326
- DOI: 10.1126/sciadv.abn5091
A clicking confinement strategy to fabricate transition metal single-atom sites for bifunctional oxygen electrocatalysis
Abstract
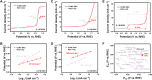
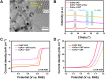
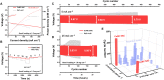
Rechargeable zinc-air batteries call for high-performance bifunctional oxygen electrocatalysts. Transition metal single-atom catalysts constitute a promising candidate considering their maximum atom efficiency and high intrinsic activity. However, the fabrication of atomically dispersed transition metal sites is highly challenging, creating a need for for new design strategies and synthesis methods. Here, a clicking confinement strategy is proposed to efficiently predisperse transitional metal atoms in a precursor directed by click chemistry and ensure successful construction of abundant single-atom sites. Concretely, cobalt-coordinated porphyrin units are covalently clicked on the substrate for the confinement of the cobalt atoms and affording a Co-N-C electrocatalyst. The Co-N-C electrocatalyst exhibits impressive bifunctional oxygen electrocatalytic performances with an activity indicator ΔE of 0.79 V. This work extends the approach to prepare transition metal single-atom sites for efficient bifunctional oxygen electrocatalysis and inspires the methodology on precise synthesis of catalytic materials.
Figures


References
-
- Mizushima K., Jones P. C., Wiseman P. J., Goodenough J. B., LixCoO2 (0<x<−1): A new cathode material for batteries of high energy density. Mater. Res. Bull. 15, 783–789 (1980).
-
- Kim J. M., Jo A., Lee K. A., Han H. J., Kim Y. J., Kim H. Y., Lee G. R., Kim M., Park Y., Kang Y. S., Jung J., Chae K. H., Lee E., Ham H. C., Ju H., Jung Y. S., Kim J. Y., Conformation-modulated three-dimensional electrocatalysts for high-performance fuel cell electrodes. Sci. Adv. 7, eabk0919 (2021). - PMC - PubMed
-
- Manthiram A., Yu X., Wang S., Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2, 16103 (2017).
-
- Kundu D., Talaie E., Duffort V., Nazar L. F., The emerging chemistry of sodium ion batteries for electrochemical energy storage. Angew. Chem. Int. Ed. 54, 3431–3448 (2015). - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

