Case Report: Tackling Complement Hyperactivation With Eculizumab in Atypical Hemolytic Uremic Syndrome Triggered by COVID-19
- PMID: 35295324
- PMCID: PMC8920243
- DOI: 10.3389/fphar.2022.842473
Case Report: Tackling Complement Hyperactivation With Eculizumab in Atypical Hemolytic Uremic Syndrome Triggered by COVID-19
Abstract
Hemolytic uremic syndrome (HUS) is a rare life-threatening disease of unrestrained complement system dysregulation, microangiopathic hemolytic anemia, thrombocytopenia, and acute renal failure in genetically predisposed individuals. In this report, we describe two cases of SARS-CoV-2-associated HUS treated with eculizumab, a C5-blocking monoclonal antibody reported to be remarkably effective in the treatment of HUS. Detailed biochemical and genetic complement system analysis is reported, and the prompt clinical response after C5 pharmacological blockade is documented. Our report provides the rationale and supports the use of terminal complement pathway inhibition for the treatment of SARS-CoV-2-associated HUS.
Keywords: coronavirus disease 2019 (COVID-19); eculizumab; hemolytic uremic syndrome; severe acute respiratory syndrome coronavirus-2 (SARS-Cov-2); thrombotic microangiopathy.
Copyright © 2022 Leone, Imeraj, Gastoldi, Mele, Liguori, Condemi, Ruggenenti, Remuzzi and Carrara.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
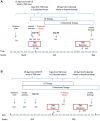
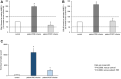
References
-
- Annane D., Heming N., Grimaldi-Bensouda L., Frémeaux-Bacchi V., Vigan M., Roux A. L., et al. (2020). Eculizumab as an Emergency Treatment for Adult Patients with Severe COVID-19 in the Intensive Care Unit: A Proof-Of-Concept Study. EClinicalMedicine 28, 100590. 10.1016/j.eclinm.2020.100590 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

