Development of a Phantom Limb Pain Model in Rats: Behavioral and Histochemical Evaluation
- PMID: 35295448
- PMCID: PMC8915728
- DOI: 10.3389/fpain.2021.675232
Development of a Phantom Limb Pain Model in Rats: Behavioral and Histochemical Evaluation
Abstract
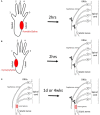
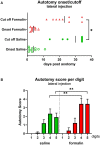
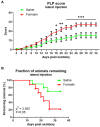
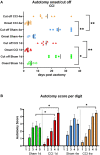
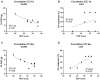
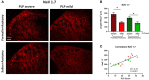
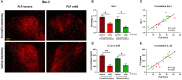
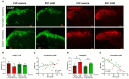
Therapeutic strategies targeting phantom limb pain (PLP) provide inadequate pain relief; therefore, a robust and clinically relevant animal model is necessary. Animal models of PLP are based on a deafferentation injury followed by autotomy behavior. Clinical studies have shown that the presence of pre-amputation pain increases the risk of developing PLP. In the current study, we used Sprague-Dawley male rats with formalin injections or constriction nerve injury at different sites or time points prior to axotomy to mimic clinical scenarios of pre-amputation inflammatory and neuropathic pain. Animals were scored daily for PLP autotomy behaviors, and several pain-related biomarkers were evaluated to discover possible underlying pathological changes. Majority displayed some degree of autotomy behavior following axotomy. Injury prior to axotomy led to more severe PLP behavior compared to animals without preceding injury. Autotomy behaviors were more directed toward the pretreatment insult origin, suggestive of pain memory. Increased levels of IL-1β in cerebrospinal fluid and enhanced microglial responses and the expression of NaV1.7 were observed in animals displaying more severe PLP outcomes. Decreased expression of GAD65/67 was consistent with greater PLP behavior. This study provides a preclinical basis for future understanding and treatment development in the management of PLP.
Keywords: GAD65/67; Nav 1.7; autotomy; axotomy; phantom limb pain.
Copyright © 2021 Jergova, Martinez, Hernandez, Schachner, Gross and Sagen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Karanikolas M, Aretha D, Tsolakis I, Monantera G, Kiekkas P, Papadoulas S, et al. . Optimized perioperative analgesia reduces chronic phantom limb pain intensity, prevalence, and frequency: a prospective, randomized, clinical trial. Anesthesiology. (2011) 114:1144–54. 10.1097/ALN.0b013e31820fc7d2 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

