Strategy for improving cell-mediated vascularized soft tissue formation in a hydrogen peroxide-triggered chemically-crosslinked hydrogel
- PMID: 35296029
- PMCID: PMC8918759
- DOI: 10.1177/20417314221084096
Strategy for improving cell-mediated vascularized soft tissue formation in a hydrogen peroxide-triggered chemically-crosslinked hydrogel
Abstract
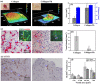
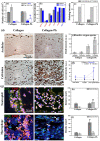
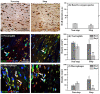
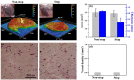
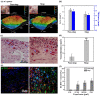
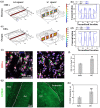
The physically-crosslinked collagen hydrogels can provide suitable microenvironments for cell-based functional vascular network formation due to their biodegradability, biocompatibility, and good diffusion properties. However, encapsulation of cells into collagen hydrogels results in extensive contraction and rapid degradation of hydrogels, an effect known from their utilization as a pre-vascularized graft in vivo. Various types of chemically-crosslinked collagen-based hydrogels have been successfully synthesized to decrease volume contraction, retard the degradation rate, and increase mechanical tunability. However, these hydrogels failed to form vascularized tissues with uniformly distributed microvessels in vivo. Here, the enzymatically chemically-crosslinked collagen-Phenolic hydrogel was used as a model to determine and overcome the difficulties in engineering vascular networks. Results showed that a longer duration of inflammation and excessive levels of hydrogen peroxide limited the capability for blood vessel forming cells-mediated vasculature formation in vivo. Lowering the unreacted amount of crosslinkers reduced the densities of infiltrating host myeloid cells by half on days 2-4 after implantation, but blood vessels remained at low density and were mainly located on the edge of the implanted constructs. Co-implantation of a designed spacer with cell-laden hydrogel maintained the structural integrity of the hydrogel and increased the degree of hypoxia in embedded cells. These effects resulted in a two-fold increase in the density of perfused blood vessels in the hydrogel. Results agreed with computer-based simulations. Collectively, our findings suggest that simultaneous reduction of the crosslinker-induced host immune response and increase in hypoxia in hydrogen peroxide-triggered chemically-crosslinked hydrogels can effectively improve the formation of cell-mediated functional vascular networks.
Keywords: Vascular tissue engineering; collagen contraction; enzymatically crosslinked; phenolic-protein hydrogels.
© The Author(s) 2022.
Conflict of interest statement
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures


Similar articles
-
Engineering large and geometrically controlled vascularized nerve tissue in collagen hydrogels to restore large-sized volumetric muscle loss.Biomaterials. 2023 Dec;303:122402. doi: 10.1016/j.biomaterials.2023.122402. Epub 2023 Nov 15. Biomaterials. 2023. PMID: 37988898 Free PMC article.
-
Bioengineering vascularized tissue constructs using an injectable cell-laden enzymatically crosslinked collagen hydrogel derived from dermal extracellular matrix.Acta Biomater. 2015 Nov;27:151-166. doi: 10.1016/j.actbio.2015.09.002. Epub 2015 Sep 5. Acta Biomater. 2015. PMID: 26348142 Free PMC article.
-
A strategy to engineer vascularized tissue constructs by optimizing and maintaining the geometry.Acta Biomater. 2022 Jan 15;138:254-272. doi: 10.1016/j.actbio.2021.11.003. Epub 2021 Nov 10. Acta Biomater. 2022. PMID: 34774782
-
Hydrogels for Engineering of Perfusable Vascular Networks.Int J Mol Sci. 2015 Jul 14;16(7):15997-6016. doi: 10.3390/ijms160715997. Int J Mol Sci. 2015. PMID: 26184185 Free PMC article. Review.
-
Recent Advances in Chemically-Modified and Hybrid Carrageenan-Based Platforms for Drug Delivery, Wound Healing, and Tissue Engineering.Polymers (Basel). 2021 May 26;13(11):1744. doi: 10.3390/polym13111744. Polymers (Basel). 2021. PMID: 34073518 Free PMC article. Review.
Cited by
-
Vascularization in Bioartificial Parenchymal Tissue: Bioink and Bioprinting Strategies.Int J Mol Sci. 2022 Aug 2;23(15):8589. doi: 10.3390/ijms23158589. Int J Mol Sci. 2022. PMID: 35955720 Free PMC article. Review.
-
Engineering large and geometrically controlled vascularized nerve tissue in collagen hydrogels to restore large-sized volumetric muscle loss.Biomaterials. 2023 Dec;303:122402. doi: 10.1016/j.biomaterials.2023.122402. Epub 2023 Nov 15. Biomaterials. 2023. PMID: 37988898 Free PMC article.
-
The Rheology and Printability of Cartilage Matrix-Only Biomaterials.Biomolecules. 2022 Jun 17;12(6):846. doi: 10.3390/biom12060846. Biomolecules. 2022. PMID: 35740971 Free PMC article.
-
Polymer-Based Hydrogels Applied in Drug Delivery: An Overview.Gels. 2023 Jun 27;9(7):523. doi: 10.3390/gels9070523. Gels. 2023. PMID: 37504402 Free PMC article. Review.
-
Reconstructing vascular networks promotes the repair of skeletal muscle following volumetric muscle loss by pre-vascularized tissue constructs.J Tissue Eng. 2023 Sep 21;14:20417314231201231. doi: 10.1177/20417314231201231. eCollection 2023 Jan-Dec. J Tissue Eng. 2023. PMID: 37744322 Free PMC article.
References
-
- Lu KG, Stultz CM. Insight into the degradation of type-I collagen fibrils by MMP-8. J Mol Biol 2013; 425: 1815–1825. - PubMed
-
- Lauer-Fields JL, Juska D, Fields GB. Matrix metalloproteinases and collagen catabolism. Biopolymers 2002; 66: 19–32. - PubMed
-
- Ferreira AM, Gentile P, Chiono V, et al.. Collagen for bone tissue regeneration. Acta Biomater 2012; 8: 3191–3200. - PubMed
LinkOut - more resources
Full Text Sources

