Insight into redox regulation of apoptosis in cancer cells with multiparametric live-cell microscopy
- PMID: 35296739
- PMCID: PMC8927414
- DOI: 10.1038/s41598-022-08509-1
Insight into redox regulation of apoptosis in cancer cells with multiparametric live-cell microscopy
Erratum in
-
Author Correction: Insight into redox regulation of apoptosis in cancer cells with multiparametric live-cell microscopy.Sci Rep. 2022 May 31;12(1):9058. doi: 10.1038/s41598-022-12901-2. Sci Rep. 2022. PMID: 35641511 Free PMC article. No abstract available.
Abstract
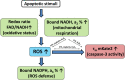
Cellular redox status and the level of reactive oxygen species (ROS) are important regulators of apoptotic potential, playing a crucial role in the growth of cancer cell and their resistance to apoptosis. However, the relationships between the redox status and ROS production during apoptosis remain poorly explored. In this study, we present an investigation on the correlations between the production of ROS, the redox ratio FAD/NAD(P)H, the proportions of the reduced nicotinamide cofactors NADH and NADPH, and caspase-3 activity in cancer cells at the level of individual cells. Two-photon excitation fluorescence lifetime imaging microscopy (FLIM) was applied to monitor simultaneously apoptosis using the genetically encoded sensor of caspase-3, mKate2-DEVD-iRFP, and the autofluorescence of redox cofactors in colorectal cancer cells upon stimulation of apoptosis with staurosporine, cisplatin or hydrogen peroxide. We found that, irrespective of the apoptotic stimulus used, ROS accumulation correlated well with both the elevated pool of mitochondrial, enzyme-bound NADH and caspase-3 activation. Meanwhile, a shift in the contribution of bound NADH could develop independently of the apoptosis, and this was observed in the case of cisplatin. An increase in the proportion of bound NADPH was detected only in staurosporine-treated cells, this likely being associated with a high level of ROS production and their resulting detoxification. The results of the study favor the discovery of new therapeutic strategies based on manipulation of the cellular redox balance, which could help improve the anti-tumor activity of drugs and overcome apoptotic resistance.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Relationship between intracellular pH, metabolic co-factors and caspase-3 activation in cancer cells during apoptosis.Biochim Biophys Acta Mol Cell Res. 2017 Mar;1864(3):604-611. doi: 10.1016/j.bbamcr.2016.12.022. Epub 2017 Jan 4. Biochim Biophys Acta Mol Cell Res. 2017. PMID: 28063999
-
Investigation of Mitochondrial Metabolic Response to Doxorubicin in Prostate Cancer Cells: An NADH, FAD and Tryptophan FLIM Assay.Sci Rep. 2017 Sep 5;7(1):10451. doi: 10.1038/s41598-017-10856-3. Sci Rep. 2017. PMID: 28874842 Free PMC article.
-
Investigating mitochondrial redox state using NADH and NADPH autofluorescence.Free Radic Biol Med. 2016 Nov;100:53-65. doi: 10.1016/j.freeradbiomed.2016.08.010. Epub 2016 Aug 9. Free Radic Biol Med. 2016. PMID: 27519271 Free PMC article. Review.
-
The Auxiliary NADH Dehydrogenase Plays a Crucial Role in Redox Homeostasis of Nicotinamide Cofactors in the Absence of the Periplasmic Oxidation System in Gluconobacter oxydans NBRC3293.Appl Environ Microbiol. 2021 Jan 4;87(2):e02155-20. doi: 10.1128/AEM.02155-20. Print 2021 Jan 4. Appl Environ Microbiol. 2021. PMID: 33127815 Free PMC article.
-
New Therapeutic Concept of NAD Redox Balance for Cisplatin Nephrotoxicity.Biomed Res Int. 2016;2016:4048390. doi: 10.1155/2016/4048390. Epub 2016 Jan 5. Biomed Res Int. 2016. PMID: 26881219 Free PMC article. Review.
Cited by
-
Assessing the impact of extracellular matrix fiber orientation on breast cancer cellular metabolism.Cancer Cell Int. 2024 Jun 5;24(1):199. doi: 10.1186/s12935-024-03385-3. Cancer Cell Int. 2024. PMID: 38840117 Free PMC article.
-
FLIM of NAD(P)H in Lymphatic Nodes Resolves T-Cell Immune Response to the Tumor.Int J Mol Sci. 2022 Dec 13;23(24):15829. doi: 10.3390/ijms232415829. Int J Mol Sci. 2022. PMID: 36555468 Free PMC article.
-
Fluorescence microscopy imaging of mitochondrial metabolism in cancer cells.Front Oncol. 2023 Jun 22;13:1152553. doi: 10.3389/fonc.2023.1152553. eCollection 2023. Front Oncol. 2023. PMID: 37427141 Free PMC article. Review.
-
The Role of Reactive Oxygen Species in Colorectal Cancer Initiation and Progression: Perspectives on Theranostic Approaches.Cancers (Basel). 2025 Feb 22;17(5):752. doi: 10.3390/cancers17050752. Cancers (Basel). 2025. PMID: 40075600 Free PMC article. Review.
-
Far-Red Fluorescent Proteins: Tools for Advancing In Vivo Imaging.Biosensors (Basel). 2024 Jul 24;14(8):359. doi: 10.3390/bios14080359. Biosensors (Basel). 2024. PMID: 39194588 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

