Implementation of an Africa-specific donor health questionnaire for human immunodeficiency virus risk screening
- PMID: 35298840
- PMCID: PMC9308630
- DOI: 10.1111/vox.13270
Implementation of an Africa-specific donor health questionnaire for human immunodeficiency virus risk screening
Abstract
Background and objectives: We had previously developed an Africa-specific donor health questionnaire (ASDHQ) based on local risk factors and designed a scoring scheme. This study assessed the performance of a new donor health questionnaire by comparing the human immunodeficiency virus (HIV) status in accepted versus deferred donors by ASDHQ and comparing the rate of risk deferrals with historical data.
Materials and methods: Data were collected during a cross-sectional study conducted over 15 months at three referral-hospital-based blood services in Cameroon. ASDHQ was administered to blood donors aged 18-65 years in the same screening conditions as the routine questionnaire. The main outcomes of the study were ASDHQ sensitivity and specificity with regard to HIV laboratory testing as well as donor deferral rates for each of the routine screening algorithms and for ASDHQ.
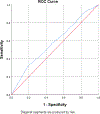
Results: Overall, 71/11,120 (0.6%) were confirmed as HIV positive. The mean ASDHQ score was 95.80 ± 4.4 in HIV-negative donors and 94.80 ± 4.4 in HIV-positive donors (p = 0.05). The optimal cut-off provided by the receiver operating characteristic (ROC) curve for the best performance of ASDHQ was 95.04. Using this optimal cut-off, the ASDHQ sensitivity and specificity were 57% and 53%, respectively (area under curve = 0.58 [0.51, 0.64], p = 0.028). Using ASDHQ, the HIV prevalence was 0.7% in deferred donors and 0.6% in accepted donors.
Conclusion: ASDHQ might be efficient only in specific conditions that maximize truthful donor responses, requiring each blood service to create an environment of trust and transparency to increase donor compliance and improve the accuracy of the questionnaire.
Keywords: Africa; HIV; blood safety; donor health questionnaire.
© 2022 International Society of Blood Transfusion.
Conflict of interest statement
Conflict of interest
The authors have disclosed no conflicts of interest.
Figures

Similar articles
-
Risk factors for human immunodeficiency virus among blood donors in Cameroon: evidence for the design of an Africa-specific donor history questionnaire.Transfusion. 2017 Aug;57(8):1912-1921. doi: 10.1111/trf.14140. Epub 2017 May 15. Transfusion. 2017. PMID: 28508402
-
Excluding blood donors at high risk of HIV infection in a west African city.BMJ. 1993 Dec 11;307(6918):1517-9. doi: 10.1136/bmj.307.6918.1517. BMJ. 1993. PMID: 8274919 Free PMC article.
-
Potential deferral criteria predictive of human immunodeficiency virus positivity among blood donors in Thailand.Transfusion. 1994 Feb;34(2):152-7. doi: 10.1046/j.1537-2995.1994.34294143945.x. Transfusion. 1994. PMID: 8310487
-
Human immunodeficiency virus-1 and hepatitis C virus RNA among South African blood donors: estimation of residual transfusion risk and yield of nucleic acid testing.Vox Sang. 2003 Jul;85(1):9-19. doi: 10.1046/j.1423-0410.2003.00311.x. Vox Sang. 2003. PMID: 12823725
-
Is there a risk of transfusion-transmissible infections after percutaneous needle treatments in blood donors? A systematic review and meta-analysis.Vox Sang. 2019 May;114(4):297-309. doi: 10.1111/vox.12780. Epub 2019 Apr 10. Vox Sang. 2019. PMID: 30972765
Cited by
-
Analysis of the most common causes of blood donor deferral in Southern Mozambique.BMC Infect Dis. 2025 Aug 7;25(1):994. doi: 10.1186/s12879-025-11393-5. BMC Infect Dis. 2025. PMID: 40775295 Free PMC article.
References
-
- World Health Organization. Current status on blood safety and availability in the WHO african region: Report of 2013 survey WHO Regional office for Africa, Brazzaville, Gongo; 2016.
-
- World Health Organization. Action framework to advance universal access to safe, effective and quality-assured blood products 2020–2023 © World Health Organization; 2020; 2020.
-
- World Health Organization. Global Safety Report on Safety and Availability WHO, Geneva, Switzerland; 2017.
-
- Lefrère J-J, Dahourouh H, Dokekias AE, Kouao MD, Diarra A, Diop S, et al. Estimate of the residual risk of transfusion-transmitted human immunodeficiency virus infection in sub-Saharan Africa: a multinational collaborative study. Transfusion (Paris) 2011. Mar;51(3):486–92. - PubMed
-
- Guekeng E, Nsagha D, Zofou D, Njunda A, Nanfack A, Fokam J, et al. Residual risk of HIV transmission through blood transfusion in five blood banks in Cameroon. Journal of Medical Research 2020;158–65.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

