Novel Pyridazin-3(2 H)-one-Based Guanidine Derivatives as Potential DNA Minor Groove Binders with Anticancer Activity
- PMID: 35300077
- PMCID: PMC8919506
- DOI: 10.1021/acsmedchemlett.1c00633
Novel Pyridazin-3(2 H)-one-Based Guanidine Derivatives as Potential DNA Minor Groove Binders with Anticancer Activity
Abstract
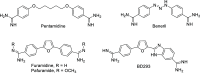
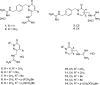
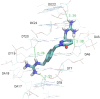
Novel aryl guanidinium analogues containing the pyridazin-3(2H)-one core were proposed as minor groove binders (MGBs) with the support of molecular docking studies. The target dicationic or monocationic compounds, which show the guanidium group at different positions of the pyridazinone moiety, were synthesized using the corresponding silyl-protected pyridazinones as key intermediates. Pyridazinone scaffolds were converted into the adequate bromoalkyl derivatives, which by reaction with N,N'-di-Boc-protected guanidine followed by acid hydrolysis provided the hydrochloride salts 1-14 in good yields. The ability of new pyridazin-3(2H)-one-based guanidines as DNA binders was studied by means of DNA UV-thermal denaturation experiments. Their antiproliferative activity was also explored in three cancer cell lines (NCI-H460, A2780, and MCF-7). Compounds 1-4 with a bis-guanidinium structure display a weak DNA binding affinity and exhibit a reasonable cellular viability inhibition percentage in the three cancer cell lines studied.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

