First Clinical Results of Fluorescence Lifetime-enhanced Tumor Imaging Using Receptor-targeted Fluorescent Probes
- PMID: 35302604
- PMCID: PMC9167767
- DOI: 10.1158/1078-0432.CCR-21-3429
First Clinical Results of Fluorescence Lifetime-enhanced Tumor Imaging Using Receptor-targeted Fluorescent Probes
Abstract
Purpose: Fluorescence molecular imaging, using cancer-targeted near infrared (NIR) fluorescent probes, offers the promise of accurate tumor delineation during surgeries and the detection of cancer specific molecular expression in vivo. However, nonspecific probe accumulation in normal tissue results in poor tumor fluorescence contrast, precluding widespread clinical adoption of novel imaging agents. Here we present the first clinical evidence that fluorescence lifetime (FLT) imaging can provide tumor specificity at the cellular level in patients systemically injected with panitumumab-IRDye800CW, an EGFR-targeted NIR fluorescent probe.
Experimental design: We performed wide-field and microscopic FLT imaging of resection specimens from patients injected with panitumumab-IRDye800CW under an FDA directed clinical trial.
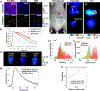
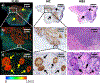
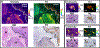
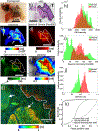
Results: We show that the FLT within EGFR-overexpressing cancer cells is significantly longer than the FLT of normal tissue, providing high sensitivity (>98%) and specificity (>98%) for tumor versus normal tissue classification, despite the presence of significant nonspecific probe accumulation. We further show microscopic evidence that the mean tissue FLT is spatially correlated (r > 0.85) with tumor-specific EGFR expression in tissue and is consistent across multiple patients. These tumor cell-specific FLT changes can be detected through thick biological tissue, allowing highly specific tumor detection and noninvasive monitoring of tumor EFGR expression in vivo.
Conclusions: Our data indicate that FLT imaging is a promising approach for enhancing tumor contrast using an antibody-targeted NIR probe with a proven safety profile in humans, suggesting a strong potential for clinical applications in image guided surgery, cancer diagnostics, and staging.
©2022 American Association for Cancer Research.
Conflict of interest statement
Figures

References
-
- van Keulen S, van den Berg NS, Nishio N, Birkeland A, Zhou Q, Lu G, Wang H-W, Middendorf L, Forouzanfar T, Martin BA, Colevas AD, and Rosenthal E, Rapid, non-invasive fluorescence margin assessment: Optical specimen mapping in oral squamous cell carcinoma. Oral oncology, 2019. 88: p. 58–65. - PMC - PubMed
-
- Gao RW, Teraphongphom N, de Boer E, van den Berg NS, Divi V, Kaplan MJ, Oberhelman NJ, Hong SS, Capes E, Colevas AD, Warram JM, and Rosenthal EL, Safety of panitumumab-IRDye800CW and cetuximab-IRDye800CW for fluorescence-guided surgical navigation in head and neck cancers. Theranostics, 2018. 8(9): p. 2488. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

