Deep brain stimulation of the thalamus restores signatures of consciousness in a nonhuman primate model
- PMID: 35302854
- PMCID: PMC8932660
- DOI: 10.1126/sciadv.abl5547
Deep brain stimulation of the thalamus restores signatures of consciousness in a nonhuman primate model
Abstract
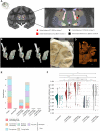
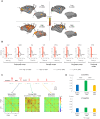
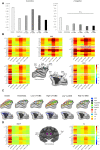
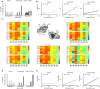
Loss of consciousness is associated with the disruption of long-range thalamocortical and corticocortical brain communication. We tested the hypothesis that deep brain stimulation (DBS) of central thalamus might restore both arousal and awareness following consciousness loss. We applied anesthesia to suppress consciousness in nonhuman primates. During anesthesia, central thalamic stimulation induced arousal in an on-off manner and increased functional magnetic resonance imaging activity in prefrontal, parietal, and cingulate cortices. Moreover, DBS restored a broad dynamic repertoire of spontaneous resting-state activity, previously described as a signature of consciousness. None of these effects were obtained during the stimulation of a control site in the ventrolateral thalamus. Last, DBS restored a broad hierarchical response to auditory violations that was disrupted under anesthesia. Thus, DBS restored the two dimensions of consciousness, arousal and conscious access, following consciousness loss, paving the way to its therapeutical translation in patients with disorders of consciousness.
Figures


References
-
- C. M. Signorelli, J. Szczotka, R. Prentner, “Explanatory profiles of models of consciousness- towards a systematic classification” (PsyArXiv, 2021);https://osf.io/f5vdu. - PMC - PubMed
-
- Dehaene S., Changeux J.-P., Experimental and theoretical approaches to conscious processing. Neuron 70, 200–227 (2011). - PubMed
-
- B. J. Baars, A Cognitive Theory of Consciousness (Cambridge Univ. Press, 1988).
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

