Soluble uric acid inhibits β2 integrin-mediated neutrophil recruitment in innate immunity
- PMID: 35303071
- PMCID: PMC11022987
- DOI: 10.1182/blood.2021011234
Soluble uric acid inhibits β2 integrin-mediated neutrophil recruitment in innate immunity
Abstract
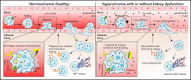
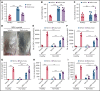
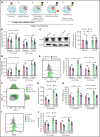
Neutrophils are key players during host defense and sterile inflammation. Neutrophil dysfunction is a characteristic feature of the acquired immunodeficiency during kidney disease. We speculated that the impaired renal clearance of the intrinsic purine metabolite soluble uric acid (sUA) may account for neutrophil dysfunction. Indeed, hyperuricemia (HU, serum UA of 9-12 mg/dL) related or unrelated to kidney dysfunction significantly diminished neutrophil adhesion and extravasation in mice with crystal- and coronavirus-related sterile inflammation using intravital microscopy and an air pouch model. This impaired neutrophil recruitment was partially reversible by depleting UA with rasburicase. We validated these findings in vitro using either neutrophils or serum from patients with kidney dysfunction-related HU with or without UA depletion, which partially normalized the defective migration of neutrophils. Mechanistically, sUA impaired β2 integrin activity and internalization/recycling by regulating intracellular pH and cytoskeletal dynamics, physiological processes that are known to alter the migratory and phagocytic capability of neutrophils. This effect was fully reversible by blocking intracellular uptake of sUA via urate transporters. In contrast, sUA had no effect on neutrophil extracellular trap formation in neutrophils from healthy subjects or patients with kidney dysfunction. Our results identify an unexpected immunoregulatory role of the intrinsic purine metabolite sUA, which contrasts the well-known immunostimulatory effects of crystalline UA. Specifically targeting UA may help to overcome certain forms of immunodeficiency, for example in kidney dysfunction, but may enhance sterile forms of inflammation.
© 2022 by The American Society of Hematology.
Figures






Comment in
-
Hyperuricemia reduces neutrophil function.Blood. 2022 Jun 9;139(23):3354-3356. doi: 10.1182/blood.2022016275. Blood. 2022. PMID: 35679075 No abstract available.
References
-
- Liew PX, Kubes P. The neutrophil's role during health and disease. Physiol Rev. 2019;99(2):1223–1248. - PubMed
-
- Németh T, Sperandio M, Mócsai A. Neutrophils as emerging therapeutic targets. Nat Rev Drug Discov. 2020;19(4):253–275. - PubMed
-
- Amulic B, Cazalet C, Hayes GL, Metzler KD, Zychlinsky A. Neutrophil function: from mechanisms to disease. Annu Rev Immunol. 2012;30(1):459–489. - PubMed
-
- Mayadas TN, Cullere X. Neutrophil beta2 integrins: moderators of life or death decisions. Trends Immunol. 2005;26(7):388–395. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

