Optimization of an O2-balanced bioartificial pancreas for type 1 diabetes using statistical design of experiment
- PMID: 35304495
- PMCID: PMC8933496
- DOI: 10.1038/s41598-022-07887-w
Optimization of an O2-balanced bioartificial pancreas for type 1 diabetes using statistical design of experiment
Abstract
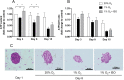
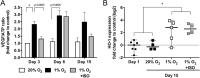
A bioartificial pancreas (BAP) encapsulating high pancreatic islets concentration is a promising alternative for type 1 diabetes therapy. However, the main limitation of this approach is O2 supply, especially until graft neovascularization. Here, we described a methodology to design an optimal O2-balanced BAP using statistical design of experiment (DoE). A full factorial DoE was first performed to screen two O2-technologies on their ability to preserve pseudo-islet viability and function under hypoxia and normoxia. Then, response surface methodology was used to define the optimal O2-carrier and islet seeding concentrations to maximize the number of viable pseudo-islets in the BAP containing an O2-generator under hypoxia. Monitoring of viability, function and maturation of neonatal pig islets for 15 days in vitro demonstrated the efficiency of the optimal O2-balanced BAP. The findings should allow the design of a more realistic BAP for humans with high islets concentration by maintaining the O2 balance in the device.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





References
-
- Orive G, et al. Engineering a clinically translatable bioartificial pancreas to treat type I diabetes. Trends Biotechnol. 2018;36:445–456. - PubMed
-
- Klymiuk N, Ludwig B, Seissler J, Reichart B, Wolf E. Current concepts of using pigs as a source for beta-cell replacement therapy of type 1 diabetes. Curr. Mol. Biol. Rep. 2016;2:73–82.
-
- Colton CK. Oxygen supply to encapsulated therapeutic cells. Adv. Drug Deliv. Rev. 2014;67–68:93–110. - PubMed
-
- Dulong J-L, Legallais C. A theoretical study of oxygen transfer including cell necrosis for the design of a bioartificial pancreas. Biotechnol. Bioeng. 2007;96:990–998. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

