Assessment of the In Vivo Relationship Between Cerebral Hypometabolism, Tau Deposition, TSPO Expression, and Synaptic Density in a Tauopathy Mouse Model: a Multi-tracer PET Study
- PMID: 35312967
- PMCID: PMC9148291
- DOI: 10.1007/s12035-022-02793-8
Assessment of the In Vivo Relationship Between Cerebral Hypometabolism, Tau Deposition, TSPO Expression, and Synaptic Density in a Tauopathy Mouse Model: a Multi-tracer PET Study
Abstract
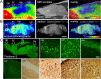
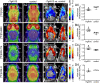
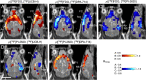
Cerebral glucose hypometabolism is a typical hallmark of Alzheimer's disease (AD), usually associated with ongoing neurodegeneration and neuronal dysfunction. However, underlying pathological processes are not fully understood and reproducibility in animal models is not well established. The aim of the present study was to investigate the regional interrelation of glucose hypometabolism measured by [18F]FDG positron emission tomography (PET) with various molecular targets of AD pathophysiology using the PET tracers [18F]PI-2620 for tau deposition, [18F]DPA-714 for TSPO expression associated with neuroinflammation, and [18F]UCB-H for synaptic density in a transgenic tauopathy mouse model. Seven-month-old rTg4510 mice (n = 8) and non-transgenic littermates (n = 8) were examined in a small animal PET scanner with the tracers listed above. Hypometabolism was observed throughout the forebrain of rTg4510 mice. Tau pathology, increased TSPO expression, and synaptic loss were co-localized in the cortex and hippocampus and correlated with hypometabolism. In the thalamus, however, hypometabolism occurred in the absence of tau-related pathology. Thus, cerebral hypometabolism was associated with two regionally distinct forms of molecular pathology: (1) characteristic neuropathology of the Alzheimer-type including synaptic degeneration and neuroinflammation co-localized with tau deposition in the cerebral cortex, and (2) pathological changes in the thalamus in the absence of other markers of AD pathophysiology, possibly reflecting downstream or remote adaptive processes which may affect functional connectivity. Our study demonstrates the feasibility of a multitracer approach to explore complex interactions of distinct AD-pathomechanisms in vivo in a small animal model. The observations demonstrate that multiple, spatially heterogeneous pathomechanisms can contribute to hypometabolism observed in AD mouse models and they motivate future longitudinal studies as well as the investigation of possibly comparable pathomechanisms in human patients.
Keywords: Alzheimer’s disease; Cerebral hypometabolism; Microglial activation; Neuroinflammation; Small animal PET; Synaptic density; Tau.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
In vivo reactive astrocyte imaging using [18F]SMBT-1 in tauopathy and familial Alzheimer's disease mouse models: A multi-tracer study.J Neurol Sci. 2024 Jul 15;462:123079. doi: 10.1016/j.jns.2024.123079. Epub 2024 Jun 4. J Neurol Sci. 2024. PMID: 38878650
-
In Vivo Visualization of Tau Accumulation, Microglial Activation, and Brain Atrophy in a Mouse Model of Tauopathy rTg4510.J Alzheimers Dis. 2018;61(3):1037-1052. doi: 10.3233/JAD-170509. J Alzheimers Dis. 2018. PMID: 29332041
-
Prodromal neuroinflammatory, cholinergic and metabolite dysfunction detected by PET and MRS in the TgF344-AD transgenic rat model of AD: a collaborative multi-modal study.Theranostics. 2021 May 3;11(14):6644-6667. doi: 10.7150/thno.56059. eCollection 2021. Theranostics. 2021. PMID: 34093845 Free PMC article.
-
Current Progress and Future Directions in Non-Alzheimer's Disease Tau PET Tracers.ACS Chem Neurosci. 2025 Jan 15;16(2):111-127. doi: 10.1021/acschemneuro.4c00319. Epub 2025 Jan 6. ACS Chem Neurosci. 2025. PMID: 39762194 Review.
-
Microglial Activation During Pathogenesis of Tauopathy in rTg4510 Mice: Implications for the Early Diagnosis of Tauopathy.J Alzheimers Dis. 2018;64(s1):S353-S359. doi: 10.3233/JAD-179933. J Alzheimers Dis. 2018. PMID: 29865054 Review.
Cited by
-
Contribution of Intravital Neuroimaging to Study Animal Models of Multiple Sclerosis.Neurotherapeutics. 2023 Jan;20(1):22-38. doi: 10.1007/s13311-022-01324-6. Epub 2023 Jan 18. Neurotherapeutics. 2023. PMID: 36653665 Free PMC article. Review.
-
Increased Cerebral Level of P2X7R in a Tauopathy Mouse Model by PET Using [18F]GSK1482160.ACS Chem Neurosci. 2024 Jun 5;15(11):2112-2120. doi: 10.1021/acschemneuro.4c00067. Epub 2024 May 22. ACS Chem Neurosci. 2024. PMID: 38776461 Free PMC article.
-
Longitudinal synaptic loss versus tau Braak staging in amnestic mild cognitive impairment.Alzheimers Dement. 2025 Feb;21(2):e14412. doi: 10.1002/alz.14412. Epub 2024 Dec 28. Alzheimers Dement. 2025. PMID: 39732507 Free PMC article.
-
SMaRT modulation of tau isoforms rescues cognitive and motor impairments in a preclinical model of tauopathy.Front Bioeng Biotechnol. 2022 Oct 5;10:951384. doi: 10.3389/fbioe.2022.951384. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36277399 Free PMC article.
-
Spreading of Tau Protein Does Not Depend on Aggregation Propensity.J Mol Neurosci. 2023 Oct;73(9-10):693-712. doi: 10.1007/s12031-023-02143-w. Epub 2023 Aug 22. J Mol Neurosci. 2023. PMID: 37606769 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

