Incidence and risk factors for hyperkalaemia in patients treated for COVID-19 with nafamostat mesylate
- PMID: 35313385
- PMCID: PMC9114925
- DOI: 10.1111/jcpt.13646
Incidence and risk factors for hyperkalaemia in patients treated for COVID-19 with nafamostat mesylate
Abstract
What is known and objective: Nafamostat mesylate (NM) is used clinically in combination with antiviral drugs to treat coronavirus disease (COVID-19). One of the adverse events of NM is hyperkalaemia due to inhibition of the amiloride-sensitive sodium channels (ENaC). The incidence and risk factors for hyperkalaemia due to NM have been studied in patients with pancreatitis but not in COVID-19. COVID-19 can be associated with hypokalaemia or hyperkalaemia, and SARS-CoV-2 is thought to inhibit ENaC. Therefore, frequency and risk factors for hyperkalaemia due to NM may differ between COVID-19 and pancreatitis. Hyperkalaemia may worsen the respiratory condition of patients. The objective of this study was to determine the incidence and risk factors for hyperkalaemia in COVID-19 patients treated with favipiravir, dexamethasone and NM.
Methods: This retrospective study reviewed the records of hospitalized COVID-19 patients treated with favipiravir and dexamethasone, with or without NM, between March 2020 and January 2021. Multivariable logistic regression analysis was performed to identify the risk factors for hyperkalaemia.
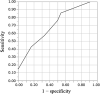
Results and discussion: Of 45 patients who received favipiravir and dexamethasone with NM for the treatment of COVID-19, 21 (47%) experienced hyperkalaemia. The duration of NM administration was a significant predictor of hyperkalaemia (odds ratio: 1.55, 95% confidence interval: 1.04-2.31, p = 0.031). The receiver-operating characteristic curve analysis determined that the cut-off value for predicting the number of days until the onset of hyperkalaemia was 6 days and the area under the curve was 0.707.
What is new and conclusion: This study revealed that the incidence of hyperkalaemia is high in patients treated for COVID-19 with NM, and that the duration of NM administration is a key risk factor. When NM is administered for the treatment of COVID-19, it should be discontinued within 6 days to minimize the risk of hyperkalaemia.
Keywords: COVID-19; amiloride-sensitive sodium channel; hyperkalaemia; nafamostat mesylate.
© 2022 John Wiley & Sons Ltd.
Conflict of interest statement
No conflicts of interest have been declared.
Figures
References
-
- World Health Organization . Coronavirus disease (COVID‐19) n.d. https://covid19.who.int Accessed December 27, 2021
-
- World Health Organization . Novel Coronavirus (2019‐nCoV) Situation Report ‐ 22 2020. https://www.who.int/docs/default‐source/coronaviruse/situation‐reports/2... Accessed September 15, 2021
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

