The accessible promoter-mediated supplementary effect of host factors provides new insight into the tropism of SARS-CoV-2
- PMID: 35313658
- PMCID: PMC8925281
- DOI: 10.1016/j.omtn.2022.03.010
The accessible promoter-mediated supplementary effect of host factors provides new insight into the tropism of SARS-CoV-2
Abstract
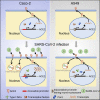
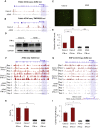
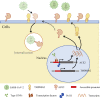
In the past year, the rapid spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) resulted in the worldwide coronavirus disease 2019 (COVID-19) pandemic. Yet our understanding of the SARS-CoV-2 tropism mechanism is still insufficient. In this study, we examined the chromatin accessibility at the promoters of host factor genes (ACE2, TMPRSS2, NRP1, BSG, CTSL, and FURIN) in 14 tissue types, 23 tumor types, and 189 cell lines. We showed that the promoters of ACE2 and TMPRSS2 were accessible in a tissue- and cell-specific pattern, which is accordant with previous clinical research on SARS-CoV-2 tropism. We were able to further verify that type I interferon (IFN) could induce angiotensin-converting enzyme 2 (ACE2) expression in Caco-2 cells by enhancing the binding of HNF1A, the transcription factor of ACE2, to ACE2 promoter without changing chromatin accessibility. We then performed transcription factor (TF)-gene interactions network and pathway analyses and discovered that the TFs regulating host factor genes are enriched in pathways associated with viral infection. Finally, we established a novel model that suggests that open chromatin at the promoter mediates the host factors' supplementary effect and ensures SARS-CoV-2 entry. Our work uncovers the relationship between epigenetic regulation and SARS-CoV-2 tropism and provides clues for further investigation of COVID-19 pathogenesis.
Keywords: ACE2; MT: Bioinformatics; SARS-CoV-2; TMPRSS2; chromatin accessibility; host factors.
© 2022 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
ACE2 and TMPRSS2 distribution in the respiratory tract of different animal species and its correlation with SARS-CoV-2 tissue tropism.Microbiol Spectr. 2024 Feb 6;12(2):e0327023. doi: 10.1128/spectrum.03270-23. Epub 2024 Jan 17. Microbiol Spectr. 2024. PMID: 38230954 Free PMC article.
-
Distinctive Roles of Furin and TMPRSS2 in SARS-CoV-2 Infectivity.J Virol. 2022 Apr 27;96(8):e0012822. doi: 10.1128/jvi.00128-22. Epub 2022 Mar 28. J Virol. 2022. PMID: 35343766 Free PMC article.
-
Prediction of the Effects of Variants and Differential Expression of Key Host Genes ACE2, TMPRSS2, and FURIN in SARS-CoV-2 Pathogenesis: An In Silico Approach.Bioinform Biol Insights. 2021 Oct 26;15:11779322211054684. doi: 10.1177/11779322211054684. eCollection 2021. Bioinform Biol Insights. 2021. PMID: 34720581 Free PMC article.
-
Contributions of human ACE2 and TMPRSS2 in determining host-pathogen interaction of COVID-19.J Genet. 2021;100(1):12. doi: 10.1007/s12041-021-01262-w. J Genet. 2021. PMID: 33707363 Free PMC article. Review.
-
Interaction of SARS-CoV-2 and Other Coronavirus With ACE (Angiotensin-Converting Enzyme)-2 as Their Main Receptor: Therapeutic Implications.Hypertension. 2020 Nov;76(5):1339-1349. doi: 10.1161/HYPERTENSIONAHA.120.15256. Epub 2020 Aug 27. Hypertension. 2020. PMID: 32851855 Free PMC article. Review.
Cited by
-
Understanding the epigenetic mechanisms in SARS CoV-2 infection and potential therapeutic approaches.Virus Res. 2022 Sep;318:198853. doi: 10.1016/j.virusres.2022.198853. Epub 2022 Jun 28. Virus Res. 2022. PMID: 35777502 Free PMC article. Review.
-
Impaired histone inheritance promotes tumor progression.Nat Commun. 2023 Jun 10;14(1):3429. doi: 10.1038/s41467-023-39185-y. Nat Commun. 2023. PMID: 37301892 Free PMC article.
References
-
- Lee E.C., Wada N.I., Grabowski M.K., Gurley E.S., Lessler J. The engines of SARS-CoV-2 spread. Science. 2020;370:406–407. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

