Human umbilical cord-derived mesenchymal stem cells alleviate insulin resistance in diet-induced obese mice via an interaction with splenocytes
- PMID: 35313972
- PMCID: PMC8935757
- DOI: 10.1186/s13287-022-02791-6
Human umbilical cord-derived mesenchymal stem cells alleviate insulin resistance in diet-induced obese mice via an interaction with splenocytes
Abstract
Background: Previous research has demonstrated that the spleen plays an important role in mesenchymal stem cell (MSC)-mediated alleviation of acute inflammation, as MSC infusion increases the spleen-derived anti-inflammatory cytokine interleukin 10 (IL-10) levels. However, studies on splenic involvement in MSC-induced protection against chronic inflammatory diseases are limited. Obesity is characterized by chronic low-grade inflammation, a key driver of insulin resistance. This study aims to evaluate the effects of MSCs on obesity-related insulin resistance and explore the underlying mechanism, particularly regarding splenic involvement.
Methods: We induced obesity in mice by feeding them high-fat diets for 20 weeks. Human umbilical cord-derived MSCs (UC-MSCs) were systemically infused into the obese mice once per week for 6 weeks. Systemic glucose metabolic homeostasis and insulin sensitivity in epididymal adipose tissue (EAT) were evaluated. Then, we conducted in vivo blockade of IL-10 during UC-MSC infusion by intraperitoneally administrating an IL-10-neutralizing antibody twice per week. We also investigated the therapeutic effects of UC-MSCs on obese mice after removal of the spleen by splenectomy.
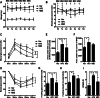
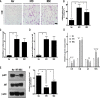
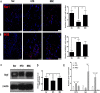
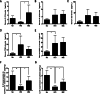
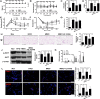
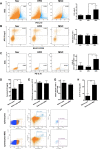
Results: UC-MSC infusions improved systemic metabolic homeostasis and alleviated insulin resistance in EAT but elicited no change in weight. Despite rare engraftment of UC-MSCs in EAT, UC-MSC infusions attenuated insulin resistance in EAT by polarizing macrophages into the M2 phenotype, coupled with elevated serum IL-10 levels. In vivo blockade of IL-10 blunted the effects of UC-MSCs on obese mice. Furthermore, UC-MSCs overwhelmingly homed to the spleen, and the ability of UC-MSCs to elevate serum IL-10 levels and alleviate insulin resistance was impaired in the absence of the spleen. Further in vivo and in vitro studies revealed that UC-MSCs promoted the capacity of regulatory T cells (Treg cells) to produce IL-10 in the spleen.
Conclusions: Our results demonstrated that UC-MSCs elevated serum IL-10 levels and subsequently promoted macrophage polarization, leading to alleviation of insulin resistance in EAT. The underlying mechanism was that UC-MSCs improved the capacity of Treg cells to produce IL-10 in the spleen. Our findings indicated that the spleen played a critical role in amplifying MSC-mediated immunomodulatory effects, which may contribute to maximizing MSC efficacy in clinical applications in the future.
Keywords: Insulin resistance; Macrophage; Mesenchymal stem cells; Obesity; Regulatory T cell; Spleen.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Decitabine assists umbilical cord-derived mesenchymal stem cells in improving glucose homeostasis by modulating macrophage polarization in type 2 diabetic mice.Stem Cell Res Ther. 2019 Aug 19;10(1):259. doi: 10.1186/s13287-019-1338-2. Stem Cell Res Ther. 2019. PMID: 31426846 Free PMC article.
-
Human umbilical cord-derived mesenchymal stem cells elicit macrophages into an anti-inflammatory phenotype to alleviate insulin resistance in type 2 diabetic rats.Stem Cells. 2016 Mar;34(3):627-39. doi: 10.1002/stem.2238. Epub 2015 Nov 17. Stem Cells. 2016. PMID: 26523620
-
The homing of human umbilical cord-derived mesenchymal stem cells and the subsequent modulation of macrophage polarization in type 2 diabetic mice.Int Immunopharmacol. 2018 Jul;60:235-245. doi: 10.1016/j.intimp.2018.04.051. Epub 2018 May 25. Int Immunopharmacol. 2018. PMID: 29778021
-
Efficacy and Safety of Umbilical Cord-Derived Mesenchymal Stromal Cell Therapy in Preclinical Models of Sepsis: A Systematic Review and Meta-analysis.Stem Cells Transl Med. 2024 Apr 15;13(4):346-361. doi: 10.1093/stcltm/szae003. Stem Cells Transl Med. 2024. PMID: 38381583 Free PMC article.
-
The therapeutic potential of stem cell-derived exosomes in the ulcerative colitis and colorectal cancer.Stem Cell Res Ther. 2022 Apr 1;13(1):138. doi: 10.1186/s13287-022-02811-5. Stem Cell Res Ther. 2022. PMID: 35365226 Free PMC article. Review.
Cited by
-
Gold Mesoporous Silica-Coated Nanoparticles for Quantifying and Qualifying Mesenchymal Stem Cell Distribution; a Proof-of-Concept Study in Large Animals.ACS Appl Bio Mater. 2025 Feb 17;8(2):1511-1523. doi: 10.1021/acsabm.4c01714. Epub 2025 Feb 3. ACS Appl Bio Mater. 2025. PMID: 39900538 Free PMC article.
-
Preclinical assessments of safety and tumorigenicity of very high doses of allogeneic human umbilical cord mesenchymal stem cells.In Vitro Cell Dev Biol Anim. 2024 Mar;60(3):307-319. doi: 10.1007/s11626-024-00852-z. Epub 2024 Feb 29. In Vitro Cell Dev Biol Anim. 2024. PMID: 38421574 Free PMC article.
-
Human umbilical cord mesenchymal stem cells in diabetes mellitus and its complications: applications and research advances.Int J Med Sci. 2023 Sep 11;20(11):1492-1507. doi: 10.7150/ijms.87472. eCollection 2023. Int J Med Sci. 2023. PMID: 37790847 Free PMC article. Review.
-
Breaking the chain in organ failure: Role of umbilical cord and bone marrow derived mesenchymal stem cells in treatment of severe acute pancreatitis.Heliyon. 2024 Aug 3;10(16):e35785. doi: 10.1016/j.heliyon.2024.e35785. eCollection 2024 Aug 30. Heliyon. 2024. PMID: 39220979 Free PMC article.
References
-
- Olefsky JM, Glass CK. Macrophages, inflammation, and insulin resistance. Annu Rev Physiol. 2010;72:219–246. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

