A pharmacogenetic interaction analysis of bevacizumab with paclitaxel in advanced breast cancer patients
- PMID: 35314692
- PMCID: PMC8938486
- DOI: 10.1038/s41523-022-00400-6
A pharmacogenetic interaction analysis of bevacizumab with paclitaxel in advanced breast cancer patients
Abstract
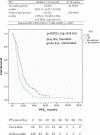
To investigate pharmacogenetic interactions among VEGF-A, VEGFR-2, IL-8, HIF-1α, EPAS-1, and TSP-1 SNPs and their role on progression-free survival (PFS) in metastatic breast cancer (MBC) patients treated with bevacizumab plus first-line paclitaxel or with paclitaxel alone. Analyses were performed on germline DNA, and SNPs were investigated by real-time PCR technique. The multifactor dimensionality reduction (MDR) methodology was applied to investigate the interaction between SNPs. The present study was an explorative, ambidirectional cohort study: 307 patients from 11 Oncology Units were evaluated retrospectively from 2009 to 2016, then followed prospectively (NCT01935102). Two hundred and fifteen patients were treated with paclitaxel and bevacizumab, whereas 92 patients with paclitaxel alone. In the bevacizumab plus paclitaxel group, the MDR software provided two pharmacogenetic interaction profiles consisting of the combination between specific VEGF-A rs833061 and VEGFR-2 rs1870377 genotypes. Median PFS for favorable genetic profile was 16.8 vs. the 10.6 months of unfavorable genetic profile (p = 0.0011). Cox proportional hazards model showed an adjusted hazard ratio of 0.64 (95% CI, 0.5-0.9; p = 0.004). Median OS for the favorable genetic profile was 39.6 vs. 28 months of unfavorable genetic profile (p = 0.0103). Cox proportional hazards model revealed an adjusted hazard ratio of 0.71 (95% CI, 0.5-1.01; p = 0.058). In the 92 patients treated with paclitaxel alone, the results showed no effect of the favorable genetic profile, as compared to the unfavorable genetic profile, either on the PFS (p = 0.509) and on the OS (p = 0.732). The pharmacogenetic statistical interaction between VEGF-A rs833061 and VEGFR-2 rs1870377 genotypes may identify a population of bevacizumab-treated patients with a better PFS.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- AIOM. Linee guida Neoplasia della mammella. https://www.aiom.it/wp-content/uploads/2020/10/2020_LG_AIOM_Neoplasie_Ma... (2020).
LinkOut - more resources
Full Text Sources
Miscellaneous

