Metabolomic profiling reveals extensive adrenal suppression due to inhaled corticosteroid therapy in asthma
- PMID: 35314841
- PMCID: PMC9350737
- DOI: 10.1038/s41591-022-01714-5
Metabolomic profiling reveals extensive adrenal suppression due to inhaled corticosteroid therapy in asthma
Erratum in
-
Author Correction: Metabolomic profiling reveals extensive adrenal suppression due to inhaled corticosteroid therapy in asthma.Nat Med. 2022 Aug;28(8):1723. doi: 10.1038/s41591-022-01949-2. Nat Med. 2022. PMID: 35859205 No abstract available.
Abstract
The application of large-scale metabolomic profiling provides new opportunities for realizing the potential of omics-based precision medicine for asthma. By leveraging data from over 14,000 individuals in four distinct cohorts, this study identifies and independently replicates 17 steroid metabolites whose levels were significantly reduced in individuals with prevalent asthma. Although steroid levels were reduced among all asthma cases regardless of medication use, the largest reductions were associated with inhaled corticosteroid (ICS) treatment, as confirmed in a 4-year low-dose ICS clinical trial. Effects of ICS treatment on steroid levels were dose dependent; however, significant reductions also occurred with low-dose ICS treatment. Using information from electronic medical records, we found that cortisol levels were substantially reduced throughout the entire 24-hour daily period in patients with asthma who were treated with ICS compared to those who were untreated and to patients without asthma. Moreover, patients with asthma who were treated with ICS showed significant increases in fatigue and anemia as compared to those without ICS treatment. Adrenal suppression in patients with asthma treated with ICS might, therefore, represent a larger public health problem than previously recognized. Regular cortisol monitoring of patients with asthma treated with ICS is needed to provide the optimal balance between minimizing adverse effects of adrenal suppression while capitalizing on the established benefits of ICS treatment.
© 2022. The Author(s), under exclusive licence to Springer Nature America, Inc.
Figures



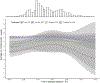
Comment in
-
Metabolomic profiling of adrenal function in asthma.Nat Rev Endocrinol. 2022 Jun;18(6):332. doi: 10.1038/s41574-022-00678-3. Nat Rev Endocrinol. 2022. PMID: 35414021 No abstract available.
-
Corticosteroid Suppression in Patients Receiving Inhaled Glucocorticoids: Time to Reassess Risk?J Clin Endocrinol Metab. 2022 Sep 28;107(10):e4256-e4258. doi: 10.1210/clinem/dgac431. J Clin Endocrinol Metab. 2022. PMID: 35908292 No abstract available.
References
-
- Global Initiative for Asthma. Global strategy for asthma management and prevention, 2021. Available at: www.ginasthma.org.
-
- Masoli M, Fabian D, Holt S & Beasley R The global burden of asthma: executive summary of the GINA Dissemination Committee report. Allergy 59, 469–478 (2004). - PubMed
-
- Becker AB & Abrams EM Asthma guidelines: the Global Initiative for Asthma in relation to national guidelines. Curr. Opin. Allergy Clin. Immunol 17, 99–103 (2017). - PubMed
-
- (2018)., C. gov. CDC - Asthma - Data and Surveillance - Asthma Surveillance Data.
-
- Greally M, Jagoe WS & Greally J The genetics of asthma. Ir. Med. J 75, 403–405 (1982). - PubMed
MeSH terms
Substances
Grants and funding
- MC_UU_00006/1/MRC_/Medical Research Council/United Kingdom
- K01 HL130629/HL/NHLBI NIH HHS/United States
- K01 HL153941/HL/NHLBI NIH HHS/United States
- R01 HL123915/HL/NHLBI NIH HHS/United States
- R01 HL155742/HL/NHLBI NIH HHS/United States
- K08 HL148178/HL/NHLBI NIH HHS/United States
- G1000143/MRC_/Medical Research Council/United Kingdom
- MR/N003284/1/MRC_/Medical Research Council/United Kingdom
- R01 HD085993/HD/NICHD NIH HHS/United States
- 14136/CRUK_/Cancer Research UK/United Kingdom
- L40 AI138248/AI/NIAID NIH HHS/United States
- R01 HL152244/HL/NHLBI NIH HHS/United States
- MC-UU_12015/1/MRC_/Medical Research Council/United Kingdom
- K99 HL159234/HL/NHLBI NIH HHS/United States
- R01 HL141826/HL/NHLBI NIH HHS/United States
- C864/A14136/CRUK_/Cancer Research UK/United Kingdom
- R01 HL139634/HL/NHLBI NIH HHS/United States
- MC_UU_12015/1/MRC_/Medical Research Council/United Kingdom
- K23 AI130408/AI/NIAID NIH HHS/United States
- G0401527/MRC_/Medical Research Council/United Kingdom
- MR/L00002/1/MRC_/Medical Research Council/United Kingdom
- U01 HG008685/HG/NHGRI NIH HHS/United States
- K23 HL151819/HL/NHLBI NIH HHS/United States
- P01 HL132825/HL/NHLBI NIH HHS/United States
- K01 HL146980/HL/NHLBI NIH HHS/United States

