Machine learning-aided design of composite mycotoxin detoxifier material for animal feed
- PMID: 35318362
- PMCID: PMC8941095
- DOI: 10.1038/s41598-022-08410-x
Machine learning-aided design of composite mycotoxin detoxifier material for animal feed
Abstract
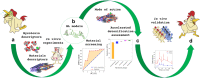
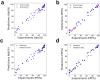
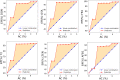
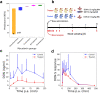
The development of food and feed additives involves the design of materials with specific properties that enable the desired function while minimizing the adverse effects related with their interference with the concurrent complex biochemistry of the living organisms. Often, the development process is heavily dependent on costly and time-consuming in vitro and in vivo experiments. Herein, we present an approach to design clay-based composite materials for mycotoxin removal from animal feed. The approach can accommodate various material compositions and different toxin molecules. With application of machine learning trained on in vitro results of mycotoxin adsorption-desorption in the gastrointestinal tract, we have searched the space of possible composite material compositions to identify formulations with high removal capacity and gaining insights into their mode of action. An in vivo toxicokinetic study, based on the detection of biomarkers for mycotoxin-exposure in broilers, validated our findings by observing a significant reduction in systemic exposure to the challenging to be removed mycotoxin, i.e., deoxynivalenol (DON), when the optimal detoxifier is administrated to the animals. A mean reduction of 32% in the area under the plasma concentration-time curve of DON-sulphate was seen in the DON + detoxifier group compared to the DON group (P = 0.010).
© 2022. The Author(s).
Conflict of interest statement
G.L.D. and V.C. are employed by Tolsa Group, which is commercializing one of the high performing composite materials identified in this study under the market name ANS. S.C. and M.H. declare no potential conflict of interest.
Figures

References
-
- Vila-Donat P, Marín S, Sanchis V, Ramos AJ. A review of the mycotoxin adsorbing agents, with an emphasis on their multi-binding capacity, for animal feed decontamination. Food Chem. Toxicol. 2018;114:246–259. - PubMed
-
- Aiko V, Mehta A. Occurrence, detection and detoxification of mycotoxins. J. Biosci. 2015;40:943–954. - PubMed
-
- Çelik K. The efficacy of mycotoxin-detoxifying and biotransforming agents in animal nutrition. Nanomycotoxicol. Treat. Mycotoxins Nano W. 2019
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

